Market Trends of Biologics CDMO Industry
Mammalian Type Segment is Expected to Hold Significant Market Share
- Biopharmaceuticals or biologics has been a growing sector, with cultured mammalian cells gaining momentum across expression systems for production due to their ability to complete the posttranslational modifications required for drug safety and efficacy.
- Mammalian cells are widely used to produce vaccines through viral infection and therapeutic proteins through genetic engineering. The allied mammalian expression systems present expansive advantages for certain types of proteins, such as trans-membrane, membrane-bound, and glycoproteins.
- Also, the Chinese hamster ovary (CHO) cells have been instrumental in manufacturing recombinant proteins with complex structures. Approximately 60% of biopharmaceuticals in clinical trials or approved by the US FDA across the last few years are therapeutic proteins, especially antibodies for treating cancers and autoimmune diseases, produced using mammalian cells. The applications of mammalian cells, thus, make it a pervasive and mass-adopted method across biologics.
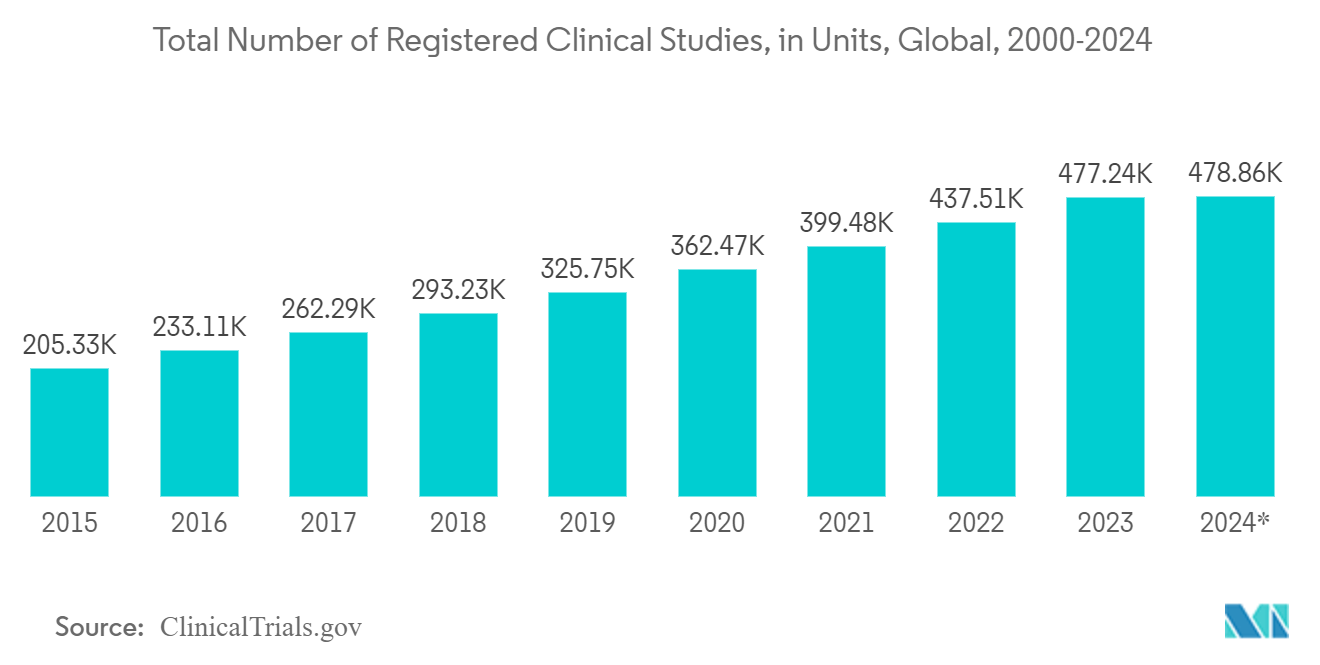
- According to ClinicalTrials.gov, Clinical studies are essential to drug development globally. The number of registered clinical trials has increased significantly in recent years. As of January 17, 2024, nearly 479 thousand clinical studies were registered globally. The number of clinical studies has increased dramatically. There were just 2,119 registered in 2000. Clinical trials have grown more complex in recent years and remain vital for researching and developing new drugs and products.
- Mammalian biologics have evolved as the preferred production technology due to pipeline increases and improvements in manufacturing technology in both volumetric and specific productivities. Also, the primary driver of consumption has been the growing incidence rate of oncology and immunological disorders.
- As biologics production by mammalian cells continues to increase, the biopharmaceutical industry is expected to continue developing next-generation-based efficient cell culture processes. Therefore, market leaders have witnessed increasing mammalian-based production capacities on the manufacturing front.
- In February 2024, leading biopharmaceutical CDMO Avid Bioservices collaborated with CRB on the design to increase the production capacity at its current Myford site in Tustin, California. To meet the market's growing demand for mammalian cell culture production of clinical and commercial biologics, Myford expanded its drug substance line (DS 2) to augment it. This reaffirmed the client's commitment to being a reliable partner for delivering innovative and high-quality biopharmaceuticals.
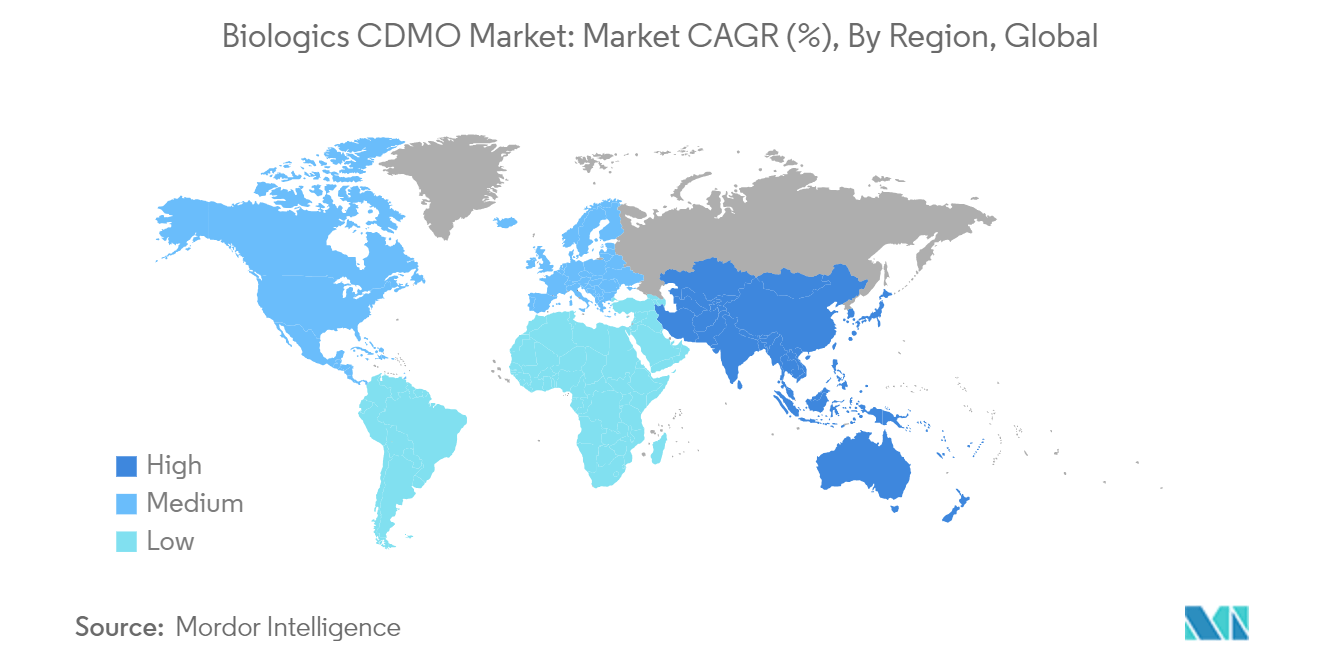
North America to Hold Major Market Share
- North America is one of the major markets for the biologics CDMO industry, owing to the presence of two major economies, such as the United States and Canada. The United States is home to one of the major pharmaceutical industries in the world and commands a significant share of the industry revenue.
- According to the study by the IQVIA Institute for Human Data Science, global medicine spending will reach USD1.8 trillion in 2026, including spending on COVID-19 vaccines. In addition to this, the region holds a prominent share of the CDMO market as well. According to Results Healthcare, the region holds about 37% of the CDMO market share and is expected to witness growth in mid-single-digit percentage points over the coming years.
- The pharmaceutical industry is one of the most innovative sectors in Canada. Pharmaceuticals, a key sector of the Canadian economy, is supported by the Canadian government, which provides a business-friendly environment for pharmaceutical companies and can leverage assets for short- and long-term business strategies.
- In the wake of the patent cliff, pharmaceutical companies in this region are reorganizing and looking for new business models built on third-party partnerships and external networks. This business model mostly relies on outsourcing most of the operations, including manufacturing, and providing growth opportunities for CDMOs in this region. Canada also holds the advantage of having a skilled workforce.
- Although Canada is going to face intense competition from emerging Asian countries, the complex manufacturing processes required for certain drugs, which need skilled personnel, a stable political environment, and the proximity to the end market of the United States and Canada, are expected to drive the growth of the biologics CDMO market in this region.
- Contract Service Providers (CSPs) in Canada are better replacements due to complex manufacturing processes for certain products, as well as the proximity to the end market in Canada and the United States. With intensifying international competition, CSPs based in Canada will seek to differentiate themselves in terms of quality and advantages of scale in their services to appeal to the demands of global leaders in pharmacy. Moreover, the growing expansions are further boosting the market growth rate in the region.