Market Size of China In Vitro Diagnostics Industry
| Study Period | 2021-2029 |
| Base Year For Estimation | 2023 |
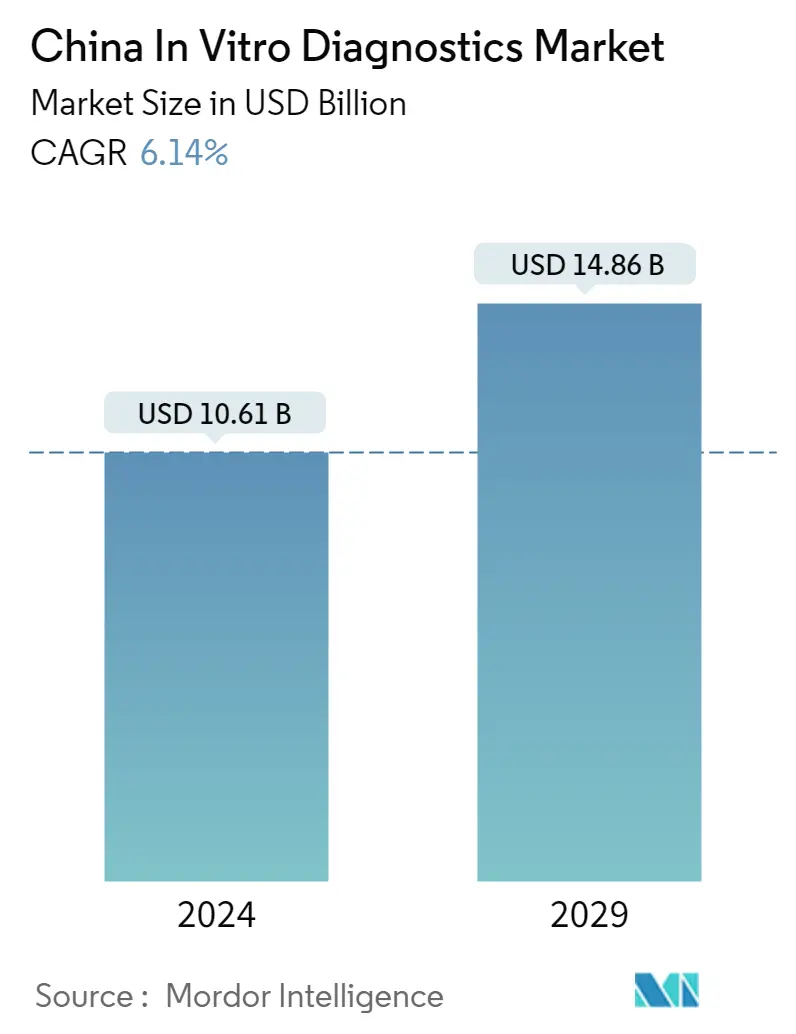
| Market Size (2024) | USD 10.61 Billion |
| Market Size (2029) | USD 14.86 Billion |
| CAGR (2024 - 2029) | 6.14 % |
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
China In-vitro Diagnostics (IVD) Market Analysis
The China In Vitro Diagnostics Market size is estimated at USD 10.61 billion in 2024, and is expected to reach USD 14.86 billion by 2029, growing at a CAGR of 6.14% during the forecast period (2024-2029).
The COVID-19 pandemic turned the spotlight on in vitro diagnostics since there was an increasing demand for IVD kits and reagents for the rapid and accurate diagnosis of SARS-CoV2 virus infection among the global population. The outbreak of COVID-19 positively impacted the market, as in vitro diagnostics involved the testing of various biological samples. This aided in the diagnosis of infectious diseases such as COVID-19. Testing remained a crucial step in controlling the COVID-19 pandemic. For instance, according to the WHO, 102,762 COVID-19 cases were recorded as of March 2021, with 4,851 deaths in China.
The increasing burden of the COVID-19 pandemic led to a high requirement for tests, which boosted the IVD market in the country. However, the stabilizing cases of COVID-19 in China were expected to stabilize the growth of the market. For instance, as per the National Health Commission (NHC), in December 2022, China reported 36,061 new COVID-19 infections, of which 4,150 were symptomatic and 31,911 were asymptomatic. Thus, the stabilizing cases of COVID-19 were expected to bring the growth of China's in vitro diagnostics market back to its pre-pandemic level.
In vitro diagnostics (IVDs) play an important role in detecting infectious diseases. A large number of molecular assays and immunoassays are being used for the detection of COVID-19. China represents one of the largest in vitro diagnostics markets in the Asia-Pacific region, and this trend is expected to continue over the forecast period. Due to an increase in the geriatric population and the high burden of chronic and infectious diseases, like diabetes, cancer, and COVID-19, the market studied is slated to show significant growth, as these chronic disorders can be diagnosed and monitored using IVD products.
In May 2021, the State Council issued a guideline to implement the country's Healthy China initiative to diminish the incidence of disability among elderly people aged between 65 and 74 years by 2022 by offering medical and health services. Such initiatives by the government are expected to boost market growth during the forecast period. But a lack of proper reimbursement and strict regulations could slow the growth of the market.
Also, the increasing number of product launches with advanced features is expected to drive the market. For instance, in September 2023, Shanghai Kehua Bio-engineering Co., Ltd has obtained the IVDR CE certificate of Class D high-risk rapid diagnostic product for HIV(1+2) Antibody (Colloidal Gold) V2.
Due to these and other factors, the in vitro diagnostic market in China is expected to grow over the next few years.
China In-vitro Diagnostics (IVD) Industry Segmentation
As per the scope of this report, in vitro diagnostics involves medical devices and consumables that are utilized to perform in vitro tests on various biological samples. They are used to diagnose various medical conditions, such as chronic diseases. The China in vitro diagnostics market is segmented by test type (clinical chemistry, molecular diagnostics, immunodiagnostics, hematology, and other types), product (instruments, reagents, and other products), usability (disposable IVD devices and reusable IVD devices), application (infectious disease, diabetes, cancer/oncology, cardiology, autoimmune disease, nephrology, and other applications), and end user (diagnostic laboratories, hospitals, and clinics, and other end users). The report offers the value in USD for the above segments.
| By Test Type | |
| Clinical Chemistry | |
| Molecular Diagnostics | |
| Immunodiagnostics | |
| Hematology | |
| Other Types |
| By Product | |
| Instrument | |
| Reagent | |
| Other Products |
| By Usability | |
| Disposable IVD Devices | |
| Reusable IVD Devices |
| By Application | |
| Infectious Disease | |
| Diabetes | |
| Cancer/Oncology | |
| Cardiology | |
| Autoimmune Disease | |
| Nephrology | |
| Other Applications |
| By End-User | |
| Diagnostic Laboratories | |
| Hospitals and Clinics | |
| Other End-Users |
China In Vitro Diagnostics Market Size Summary
The China in vitro diagnostics market is poised for significant growth over the forecast period, driven by the increasing demand for diagnostic tests and the rising prevalence of chronic and infectious diseases. The market's expansion is largely attributed to the heightened focus on early disease detection and monitoring, particularly in the wake of the COVID-19 pandemic, which underscored the critical role of IVDs in managing public health crises. The country's robust healthcare initiatives, such as the Healthy China initiative, aim to reduce disability rates among the elderly, further propelling the demand for diagnostic services. Additionally, the introduction of advanced diagnostic products and technologies, including molecular diagnostics, is expected to enhance the market's growth trajectory.
China's in vitro diagnostics market is characterized by a competitive landscape with key players actively expanding their product offerings and leveraging strategic collaborations to strengthen their market presence. The market benefits from a high concentration of industry leaders and a supportive regulatory environment, which facilitates the launch of innovative diagnostic solutions. The increasing incidence of cancer and other chronic conditions is a major driver of market demand, as early detection and intervention are crucial for improving patient outcomes. Furthermore, the establishment of technical innovation centers and the introduction of new guidelines for medical devices underscore the government's commitment to advancing the diagnostics sector, ensuring sustained market growth in the coming years.
China In Vitro Diagnostics Market Size - Table of Contents
-
1. MARKET DYNAMICS
-
1.1 Market Overview
-
1.2 Market Drivers
-
1.2.1 High Burden of Chronic Diseases and Infectious Diseases
-
1.2.2 Increasing Use of Point-of-Care (POC) Diagnostics Spurring the IVD Market
-
1.2.3 Increasing Awareness and Acceptance of Personalized Medicine
-
-
1.3 Market Restraints
-
1.3.1 Lack of Proper Reimbursement
-
1.3.2 Stringent Regulatory Framework
-
-
1.4 Porter's Five Forces Analysis
-
1.4.1 Threat of New Entrants
-
1.4.2 Bargaining Power of Buyers/Consumers
-
1.4.3 Bargaining Power of Suppliers
-
1.4.4 Threat of Substitute Products
-
1.4.5 Intensity of Competitive Rivalry
-
-
-
2. MARKET SEGMENTATION (Market Size by Value - USD)
-
2.1 By Test Type
-
2.1.1 Clinical Chemistry
-
2.1.2 Molecular Diagnostics
-
2.1.3 Immunodiagnostics
-
2.1.4 Hematology
-
2.1.5 Other Types
-
-
2.2 By Product
-
2.2.1 Instrument
-
2.2.2 Reagent
-
2.2.3 Other Products
-
-
2.3 By Usability
-
2.3.1 Disposable IVD Devices
-
2.3.2 Reusable IVD Devices
-
-
2.4 By Application
-
2.4.1 Infectious Disease
-
2.4.2 Diabetes
-
2.4.3 Cancer/Oncology
-
2.4.4 Cardiology
-
2.4.5 Autoimmune Disease
-
2.4.6 Nephrology
-
2.4.7 Other Applications
-
-
2.5 By End-User
-
2.5.1 Diagnostic Laboratories
-
2.5.2 Hospitals and Clinics
-
2.5.3 Other End-Users
-
-
China In Vitro Diagnostics Market Size FAQs
How big is the China In Vitro Diagnostics Market?
The China In Vitro Diagnostics Market size is expected to reach USD 10.61 billion in 2024 and grow at a CAGR of 6.14% to reach USD 14.86 billion by 2029.
What is the current China In Vitro Diagnostics Market size?
In 2024, the China In Vitro Diagnostics Market size is expected to reach USD 10.61 billion.

