Market Trends of Dermatological Therapeutics Industry
Psoriasis Segment is Expected to Hold a Significant Market Share Over the Forecast Period
Psoriasis is a chronic autoimmune skin disease that speeds up the growth cycle of skin cells. It causes patches of thick red skin and silvery scales on the elbows, face, palms, knees, scalp, lower back, and soles of feet. The most common type of psoriasis is plaque psoriasis, and misguided T-cell attacks on the skin cause it. The rising prevalence of psoriasis, the increasing number of clinical trials for psoriasis drug development, and the rising developments by major market players are expected to boost the segment's growth.
The rising prevalence of psoriasis is a major factor driving the segment. For instance, according to the data updated by the National Psoriasis Foundation in December 2022, around 125 million people worldwide had psoriasis in 2022, almost equal to 2-3% of the total population. The source also stated that more than 8 million Americans had psoriasis in 2022. Thus, the high prevalence of psoriasis is expected to boost the adoption of dermatological therapeutics. As psoriasis is often associated with the elderly, the rising geriatric population is also expected to drive segmental growth.
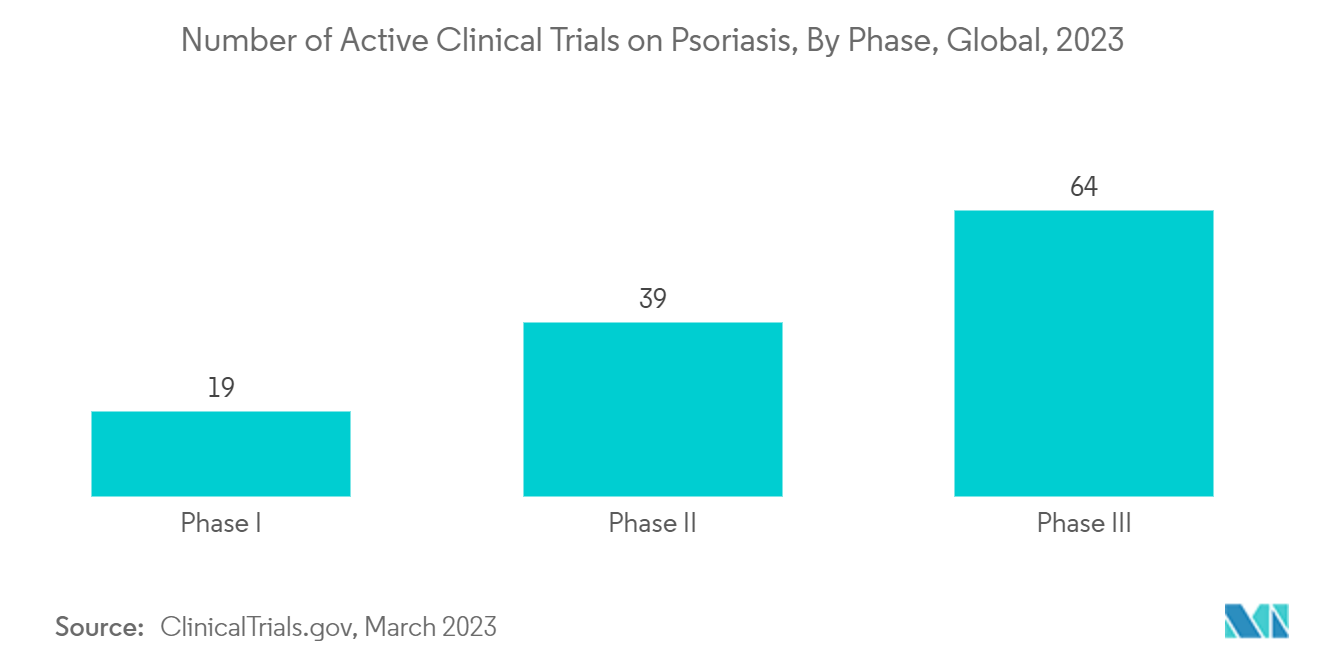
As per the data published by clinicaltrials.gov, in February 2023, globally, approximately 325 ongoing clinical trials were being conducted for the treatment of psoriasis. These are expected to receive approvals for the treatment of the disease in the future, thus driving the segment's growth.
The new product launches, the focus of market players in expanding their market for dermatology therapeutics, and collaborations and acquisitions are anticipated to boost the segment. For instance, in December 2021, the US FDA approved the expanded use of Amgen Inc.'s drug Otezla to treat adults with mild to moderate plaque psoriasis.
Thus, due to factors such as the rising prevalence of psoriasis, the rising geriatric population, the increasing developments by key players, and the rising number of psoriasis clinical trials, the segment is expected to witness steady growth over the forecast period.
North America is Expected to Have a Significant Market Share Over the Forecast Period
North America is expected to hold a significant market share due to increasing healthcare expenditure, growing awareness about skin diseases, and the strong presence of pharmaceutical companies.
The rising prevalence of dermatological diseases is a major factor that is expected to boost the market in the region. For instance, according to the data published by the Asthma and Allergy Foundation of America in August 2022, around 16.5 million adults had atopic dermatitis (AD), with 6.6 million reporting moderate-to-severe symptoms in 2022. Thus, the high prevalence of such dermatological diseases is expected to boost the adoption of dermatological therapeutics in the region.
The rising geriatric population is also a major driver of the market. For instance, according to the data published by Statistics Canada in July 2022, around 7,330,605 people are 65 years of age or older in Canada, accounting for 18.8% of the total population.
The new product launches by key regional market players are also anticipated to boost the market's growth. For instance, in July 2021, Sol-Gel Technologies Ltd announced that the FDA approved its first proprietary drug product, TWYNEO (tretinoin/benzoyl peroxide) cream, 0.1%/3%, indicated for the treatment of acne vulgaris in adults and pediatric patients nine years of age and older. Similarly, in September 2021, Incyte announced that the US FDA approved Opzelura (ruxolitinib) cream for the short-term and non-continuous chronic treatment of mild to moderate atopic dermatitis (AD) in non-immunocompromised patients 12 years of age and older whose disease is not adequately controlled with topical prescription therapies, or when those therapies are not advisable. Thus, the rising product launches by key market players are mainly driving the North American market.
Thus, due to factors such as the rising prevalence of dermatological diseases, the rising geriatric population, and the increasing developments by key market players, the market is expected to witness steady growth in the region over the forecast period.