Market Trends of Duchenne Muscular Dystrophy Treatment Industry
The Exon-skipping Segment is Expected to Hold a Major Market Share Over the Forecast Period
Mutations involving internal deletions in the gene responsible for encoding dystrophin, a protein vital for maintaining muscle cell membrane integrity, are the predominant drivers of Duchenne muscular dystrophy. A promising therapeutic avenue involves the strategic masking of an exon located proximal to the deleted regions, facilitating the alignment of the remaining exons. This approach, known as exon-skipping, is one of the most efficacious treatment modalities for reinstating the production of a truncated yet functional dystrophin protein.
Market players focus on frequent launches in exon skipping technology, a significant factor driving the segment’s growth. For instance, in November 2023, six therapies were approved in Europe for treating DMD, including four exon-skipping drugs (eteplirsen, viltolarsen, golodiresen, and casimersen). These drugs require lifetime treatment and work to trick muscle cells into generating a slightly shorter but functional dystrophin protein. This approach bypasses the effect of the disease-causing mutation. Similarly, in January 2024, Sarepta Therapeutics Inc. reported positive data from Part B of the MOMENTUM study (Study SRP-5051-201), a global, Phase 2, multi-ascending dose clinical trial of SRP-5051 (vesleteplirsen) that enrolled patients aged 8-21 years. SRP-5051 represents a cutting-edge peptide phosphorodiamidate morpholino oligomer (PPMO) therapy designed for individuals afflicted with DMD who exhibit exon 51 susceptibility. Consequently, substantial segment expansion is anticipated throughout the projected period, owing to product introductions, regulatory endorsements, and technological innovations.
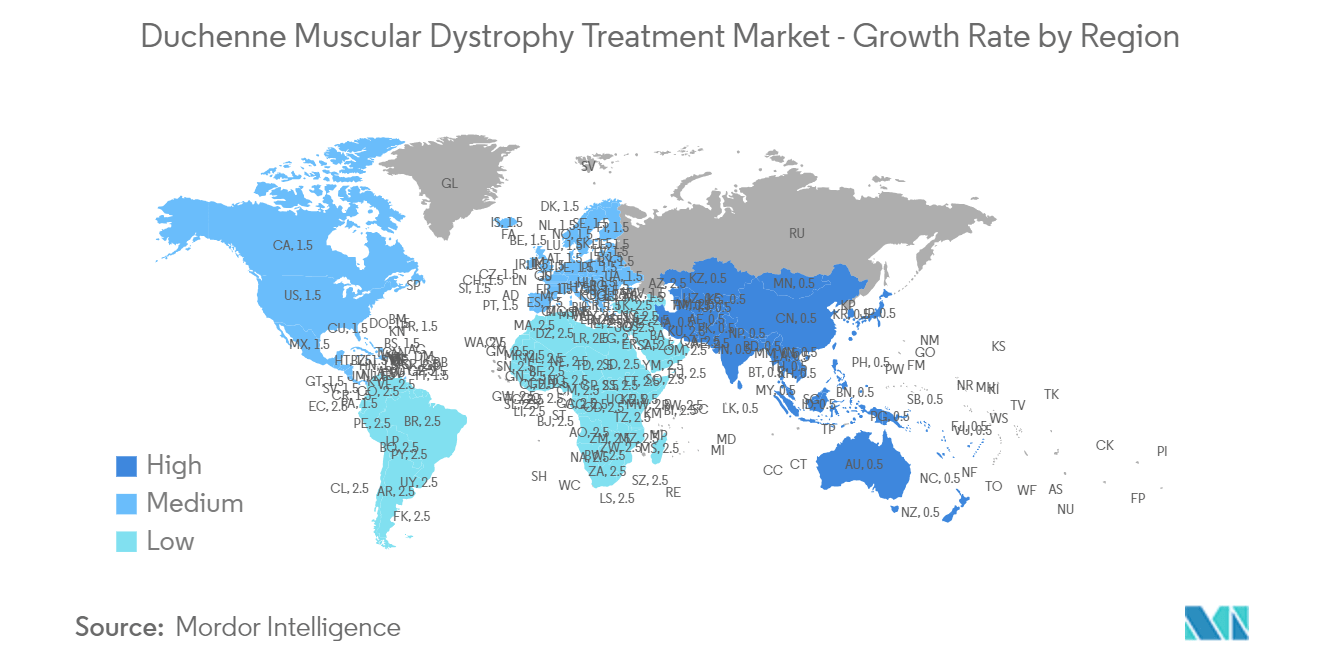
North America is Expected to Witness Considerable Growth Over the Forecast Period
North America is expected to grow considerably due to new product innovations, high healthcare expenditure, and government awareness programs. Due to the increased disease prevalence and anticipated launches of attractive pipeline candidates, the United States has led the regional market and is expected to keep this position. The market is poised for growth with the increasing number of clinical trials conducted worldwide, notably within the United States. For instance, in June 2023, Sarepta Therapeutics Inc. obtained accelerated approval from the US Food and Drug Administration (FDA) for ELEVIDYS (delandistrogene moxeparvovec-rokl), an adeno-associated virus-based gene therapy. This therapy targets the treatment of ambulatory pediatric patients aged 4-5 years diagnosed with DMD and confirmed mutations in the DMD gene. Similarly, in March 2024, Catalyst Pharmaceuticals Inc. announced the US commercial launch of AGAMREE (vamorolone) oral suspension 40 mg/mL for treating Duchenne muscular dystrophy (DMD) in patients aged two years and older.
The Duchenne muscular dystrophy treatment market is expected to grow significantly in the region due to huge unmet needs and favorable initiatives by key players. For instance, in May 2023, PepGen Inc. received a No Objection Letter (NOL) for its Clinical Trial Application (CTA) from Health Canada for its Phase 2 CONNECT1-EDO51 study to initiate an open-label, multiple ascending dose (MAD) clinical trial of PGN-EDO51 in patients with Duchenne muscular dystrophy (DMD) amenable to an exon 51 skipping approach. The presence of several new medications in the pipeline, the availability of gene therapies, and the anticipated increase in treatment rates will be significant growth drivers for the DMD market. Thus, significant market growth is expected over the forecast period in North America.