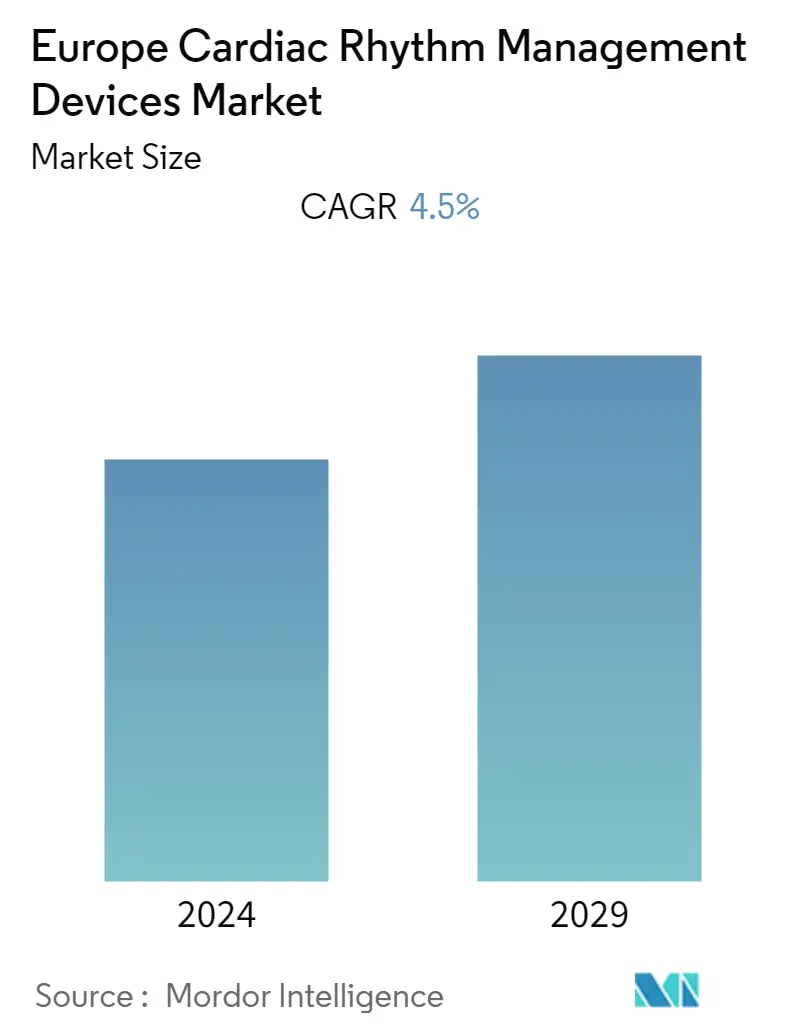
Europe Cardiac Rhythm Management Devices Market Size
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
| Historical Data Period | 2019 - 2022 |
| CAGR | 4.50 % |
| Market Concentration | Low |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Europe Cardiac Rhythm Management Devices Market Analysis
The European cardiac rhythm management devices market is expected to register a CAGR of 4.5% during the forecast period.
The emergence of the COVID-19 pandemic led to the lockdown restrictions in the region that affected the diagnosis and monitoring of cardiovascular diseases and the logistics and supply chain of medical devices, including cardiac rhythm management devices, which affected the market growth. For instance, as per the article published in BMJ journal in August 2021, the number of imaging diagnostic procedures for cardiovascular disorders decreased by 45% in the early stages of the pandemic and by 69% in the later stages of the pandemic in the European region.
However, relaxed lockdowns and the resumption of healthcare practices are expected to accelerate market growth in the post-pandemic scenario. For instance, the revenue of the Cardiac Rhythm and Heart Failure segment of Medtronic increased from USD 5,584 million in 2021 to USD 5,908 million in 2022. Also, people with underlying cardiovascular conditions are more prone to severe COVID-19 and long-term risks, such as stroke, arrhythmias, and myocarditis, among others. Thus, the demand for CRM devices is expected to grow in the post-pandemic period.
The market has shown consistent growth owing to the increasing prevalence of cardiovascular disorders, technological advancements, and the rise in the use of ambulatory and home services for cardiac monitoring. The increasing prevalence of cardiovascular disorders is one of the major factors driving the cardiac rhythm management devices market. For instance, as per the BHF 2022 report, vascular dementia affected at least 150,000 people in the United Kingdom in 2022, and the number is predicted to rise to 350,000 by 2030. Hence, the high prevalence of cardiac diseases in European countries is anticipated to be the driving factor for the cardiac rhythm management devices market. Risk factors include smoking, obesity, lack of exercise, high cholesterol, high blood pressure, and diabetes.
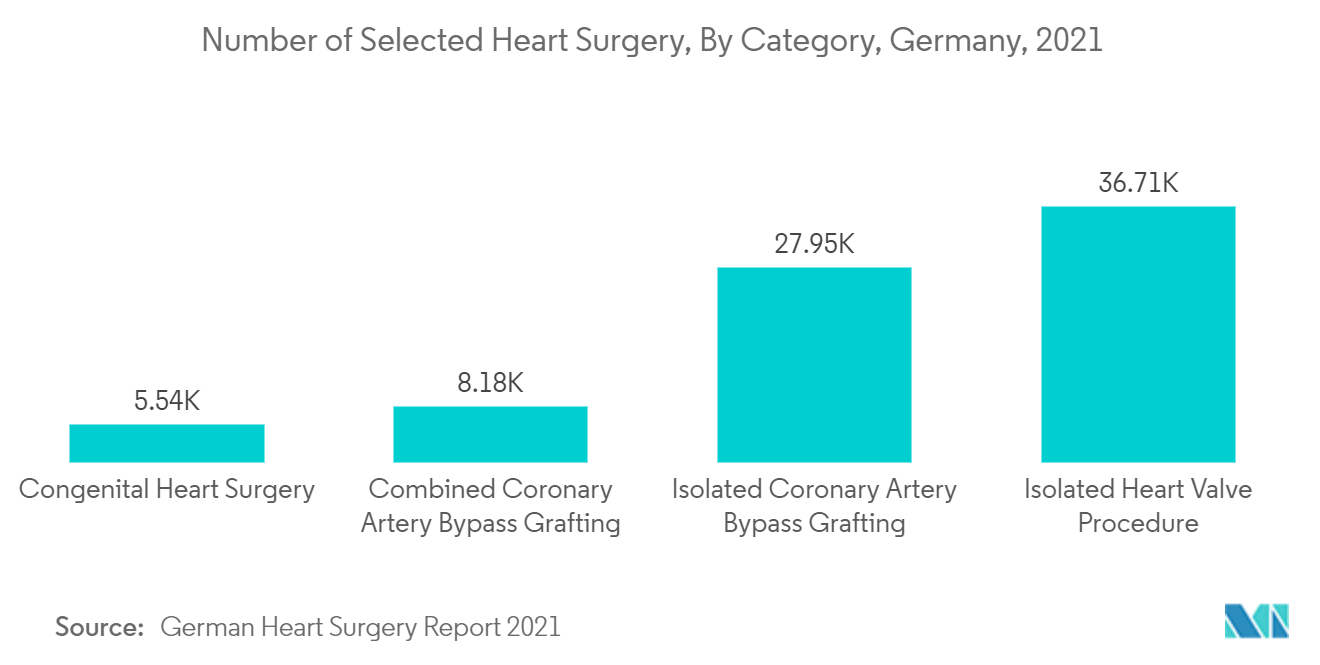
Additionally, the German Heart Surgery Report 2021 stated that considering the range of heart operations per 100,000 inhabitants showed a minimum of 96.3 (Baden-Württemberg, population: 11,123,393) and a maximum of 156.1 (Sachsen-Anhalt, population: 2,172,221), while the nationwide mean value at the end of 2021 was 109.9. Thus, the increasing cases of cardiovascular diseases across Europe are driving the growth of the market.
Due to technological innovations and product launches, cardiac rhythm management devices are anticipated to witness strong growth in the study period. The adoption of cardiac rhythm management devices is likely to increase in hospital and home care settings. For instance, in April 2021, Colruyt Group established that more than 60 shops of Spar Colruyt Group in Belgium were anticipated to install automatic external defibrillator devices after successful crowdfunding with the motto 'Your Spar has a heart for the neighborhood.' Additionally, with the advancement in heart rhythm management technology, in April 2022, Boston expanded its digital offering with the European launch of Heart Connect. It is among the newest tools in a growing market for remote management and support in heart rhythm management. Hence, considering technological advancements and the adoption of home-based cardiac monitoring care, the market is anticipated to grow in the forecast period. However, the high cost of cardiovascular devices is likely to restrain the market growth.
Europe Cardiac Rhythm Management Devices Market Trends
This section covers the major market trends shaping the Europe Cardiac Rhythm Management Devices Market according to our research experts:
External Defibrillator (ED) Segment is Expected to Hold a Significant Share During the Forecast Period
An external defibrillator is a device used in case of an emergency cardiac arrest. It is a portable device strategically placed, often in wall cabinets, in locations where large numbers of people gather, and even hospitals keep an external defibrillator or multiple external defibrillators. These external defibrillators give necessary defibrillation (restarting of the heart) treatment within the first critical minutes. There are two types of automated external defibrillators: semi-automated external defibrillators and fully automated external defibrillators. The external defibrillator segment is likely to witness growth during the forecast period owing to factors such as the increasing prevalence of cardiac diseases in Europe and technological advancements in the cardiology field. An external defibrillator is frequently used to treat arrhythmia or irregular heartbeats because the device sends an electric pulse or shock to the heart to restore a normal heartbeat.
A fully automated external defibrillator (AED) automatically delivers a safe, effective shock and can reduce extended delays associated with hesitating to press the shock button during a rescue. R&D is conducted in the European region to demonstrate the efficiency of AED. For instance, as per the article published on NLM in 2021, to increase the rate of defibrillation before the arrival of the emergency medical service (EMS), implementing first responders (FR) systems including both citizens trained in cardiopulmonary resuscitation (CPR) and medical professional equipped with an AED is helpful. Hence such studies and initiatives boost the awareness and importance of the utilization of external defibrillators, thereby fuelling the market growth.
Furthermore, the presence of advanced healthcare infrastructure and a continuous rise in the prevalence of cardiovascular cases in the country is expected to boost the market. For instance, as per BHF 2022 report, in England, around 6.4 million people were living with heart and circulatory diseases in 2021. Additionally, according to the German Heart Surgery Report 2021, 49.5% of heart procedures were performed on patients between the ages of 20 and 69. Thus, all these factors are increasing the demand for external defibrillators, driving the market's growth. Additionally, the presence of competitors, government initiatives, and cardiology awareness programs in the field of the external defibrillator segment is likely to boost the market growth. These factors can increase the chance of the adoption of external defibrillator devices. For instance, in June 2021, the Italian Resuscitation Council's proposals to improve Italy's ability to save lives from sudden cardiac arrest became law. As per this law, AEDs are mandatory in all government buildings, infrastructure, and public transport. Therefore, such government initiatives relating to AED are resulting in the adoption of external defibrillators, which is likely to fuel market growth.
Considering the above-mentioned factors, the external defibrillator segment is likely to witness growth during the forecast period.
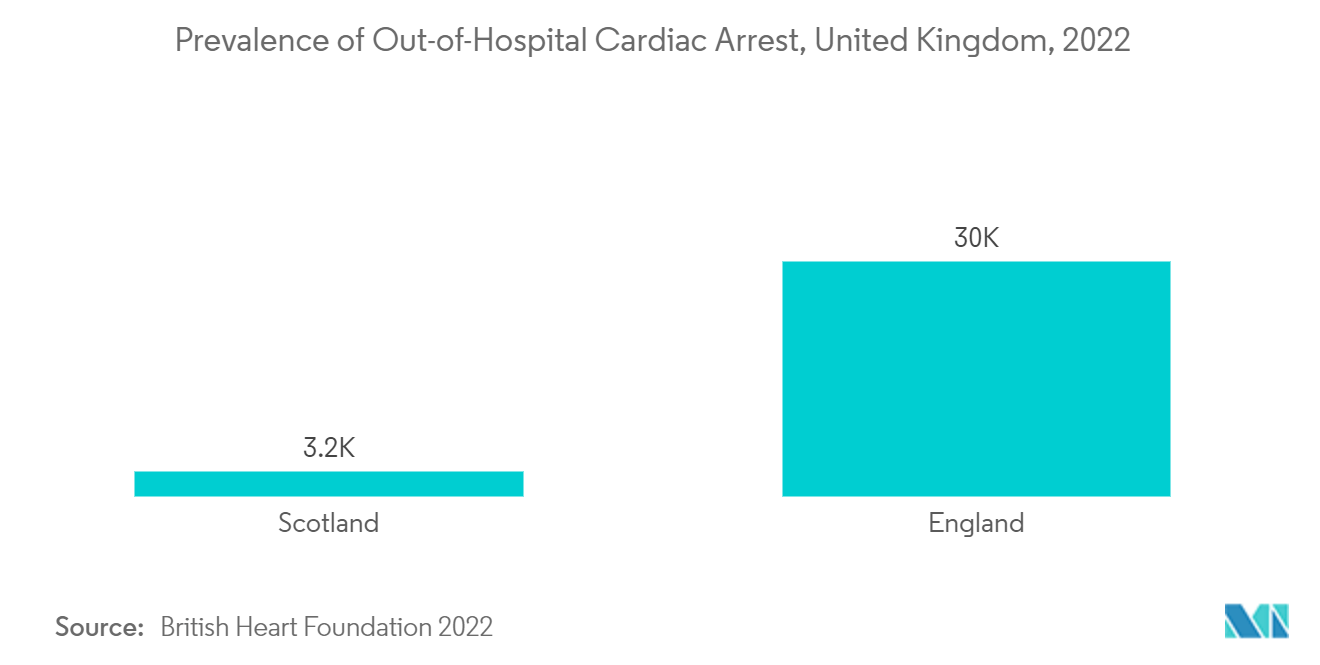
United Kingdom is Likely to Witness a Growth in the Market During the Forecast Period
The United Kingdom is expected to have a significant market share in the cardiac rhythm management devices market due to the developed healthcare infrastructure, government initiatives, and the rising burden of cardiovascular diseases. For instance, as per the BHF 2022 report, around 7.6 million people are living with heart and circulatory diseases in the United Kingdom. The same source stated that around 4 million males and 3.6 million females were living with heart and circulatory diseases in the United Kingdom in 2022. Also, around twice as many people are living with heart and circulatory diseases in the United Kingdom than with cancer and Alzheimer's disease combined. Considering the high prevalence of cardiac diseases, the demand for cardiac rhythm devices is expected to increase in the country, which is anticipated to boost market growth.
Furthermore, the presence of competitors, mergers, and product launches in the United Kingdom is likely to boost the market growth. These are expected to have a favourable effect on the market by increasing product availability and enabling more competition. For instance, in March 2022, a personal defibrillator was launched in the United Kingdom, named CellAED. Rapid Response Revival developed the defibrillator to increase access to defibrillators and overcome barriers to their use. The world's first personal, portable defibrillator uses sophisticated technology designed to improve the chances of surviving a sudden cardiac arrest.
Additionally, in February 2022, Ceryx Medical Limited collaborated with scientists at the Auckland Bioengineering Institute (ABI) and the Universities of Bath and Bristol to develop Cysoni. This bionic device paces the heart with real-time respiratory modulation. The innovation stems from the idea that heart rate increases and decreases with each breath in normal physiology, termed 'respiratory sinus arrhythmia (RSA). Owing to the abovementioned factors, the United Kingdom is likely to witness growth in the cardiac rhythm management devices market in the forecast period.
Europe Cardiac Rhythm Management Devices Industry Overview
The European cardiac rhythm management devices market is highly competitive and consists of a few major players. Companies like Abbott, Biotronik SE & Co KG, Boston Scientific Corporation, Koninklijke Philips NV, LivaNova PLC, Medtronic, SchillerAG, Shenzhen Mindray Biomedical Electronics Co. Ltd, among others, hold a substantial market share in the cardiac rhythm management devices market. Geographical expansion, collaboration, mergers and acquisitions, and new product development are key strategies of players in the cardiac rhythm management devices market.
Europe Cardiac Rhythm Management Devices Market Leaders
-
Boston Scientific Corporation
-
Biotronik SE & Co KG
-
Cardinal Health
-
Medtronic
-
Abbott
*Disclaimer: Major Players sorted in no particular order
Europe Cardiac Rhythm Management Devices Market News
- November 2022: Heart experts at University Hospital Southampton (UHS) implanted a new leadless pacemaker defibrillator system to treat patients at risk of sudden cardiac arrest. The surgery is a component of a global clinical trial involving the implantation of the innovative leadless pacemaker EMPOWER and the subcutaneously implanted cardioverter-defibrillator (S-ICD).
- February 2022: Abbott reported the patient implants of a dual-chamber leadless pacemaker system as part of its AVEIR DR i2i pivotal clinical study. The implant of Abbott's investigational Aveir dual-chamber leadless pacemaker represents a significant technological milestone for leadless pacing technology. The study aims to enroll up to 550 patients from up to 80 sites, including Europe, and after receiving an implant, every patient is likely to be monitored for at least 12 months.
Europe Cardiac Rhythm Management Devices Market Report - Table of Contents
1. INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
- 4.1 Market Overview
-
4.2 Market Drivers
- 4.2.1 Increasing Prevalence of Cardiovascular Disorders
- 4.2.2 Technological Advancements
- 4.2.3 Rise in the Use of Ambulatory and Home Services for Cardiac Monitoring
-
4.3 Market Restraints
- 4.3.1 High Cost of Devices
-
4.4 Porter's Five Force Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
-
5.1 By Product
- 5.1.1 Defibrillators
- 5.1.1.1 Implantable Cardioverter Defibrillators (ICDS)
- 5.1.1.2 External Defibrillators (ED)
- 5.1.2 Pacemakers
- 5.1.2.1 Implantable Pacemakers (ICPS)
- 5.1.2.2 External Pacemakers
- 5.1.3 Cardiac Resynchronization Therapy Devices
- 5.1.3.1 Cardiac Resynchronization Therapy Defibrillators (CRT-D)
- 5.1.3.2 Cardiac Resynchronization Therapy Pacemakers (CRT-P)
-
5.2 Geography
- 5.2.1 Germany
- 5.2.2 United Kingdom
- 5.2.3 France
- 5.2.4 Italy
- 5.2.5 Spain
- 5.2.6 Rest of Europe
6. COMPETITIVE LANDSCAPE
-
6.1 Company Profiles
- 6.1.1 Abbott
- 6.1.2 Biotronik SE & Co KG
- 6.1.3 Boston Scientific Corporation
- 6.1.4 Cardinal Health
- 6.1.5 LivaNova PLC
- 6.1.6 Medtronic
- 6.1.7 SchillerAG
- 6.1.8 Shenzhen Mindray Biomedical Electronics Co. Ltd
- 6.1.9 Zoll Medical Corporation
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
** Subject To AvailablityEurope Cardiac Rhythm Management Devices Industry Segmentation
As per the scope of the report, cardiac rhythm management is part of electrophysiology. It is used to manage the functioning of the heart. Cardiac rhythm management devices perform many functions, including bradycardia pacing, monitoring for arrhythmias, cardiac resynchronization for heart failure, defibrillation, and anti-tachycardia pacing for tachyarrhythmias. The last decade has observed an ongoing evolution in the use of cardiac rhythm management devices, including cardiac resynchronization therapy, pacemakers, implantable cardioverter defibrillators, and loop recorders. General practitioners (GP) are increasingly involved in the follow-up and management of patients with these devices.
The European cardiac rhythm management devices market is segmented by Product (Defibrillators (Implantable Cardioverter Defibrillators (ICDS) and External Defibrillators (ED)), Pacemakers (Implantable Pacemakers (ICPS) and External Pacemakers), and Cardiac Resynchronization Therapy Devices (Cardiac Resynchronization Therapy Defibrillators (CRT-D) and Cardiac Resynchronization Therapy Pacemakers (CRT-P))) and Geography (Germany, United Kingdom, France, Italy, Spain, Rest of Europe). The report offers the value in USD million for the above segments.
| By Product | Defibrillators | Implantable Cardioverter Defibrillators (ICDS) |
| External Defibrillators (ED) | ||
| By Product | Pacemakers | Implantable Pacemakers (ICPS) |
| External Pacemakers | ||
| By Product | Cardiac Resynchronization Therapy Devices | Cardiac Resynchronization Therapy Defibrillators (CRT-D) |
| Cardiac Resynchronization Therapy Pacemakers (CRT-P) | ||
| Geography | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe |
Europe Cardiac Rhythm Management Devices Market Research FAQs
What is the current Europe Cardiac Rhythm Management Devices Market size?
The Europe Cardiac Rhythm Management Devices Market is projected to register a CAGR of 4.5% during the forecast period (2024-2029)
Who are the key players in Europe Cardiac Rhythm Management Devices Market?
Boston Scientific Corporation, Biotronik SE & Co KG, Cardinal Health, Medtronic and Abbott are the major companies operating in the Europe Cardiac Rhythm Management Devices Market.
What years does this Europe Cardiac Rhythm Management Devices Market cover?
The report covers the Europe Cardiac Rhythm Management Devices Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Europe Cardiac Rhythm Management Devices Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Europe Cardiac Rhythm Management Devices Industry Report
Statistics for the 2024 Europe Cardiac Rhythm Management Devices market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Europe Cardiac Rhythm Management Devices analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.



