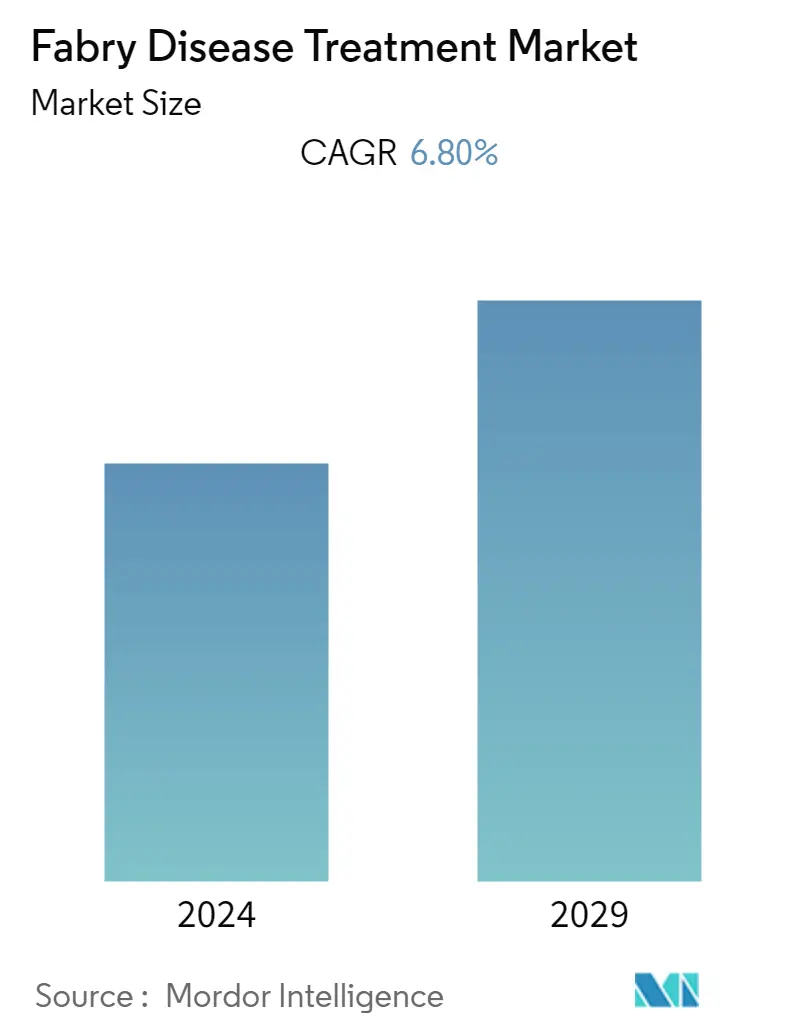
Fabry Disease Treatment Market Size
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| CAGR | 6.80 % |
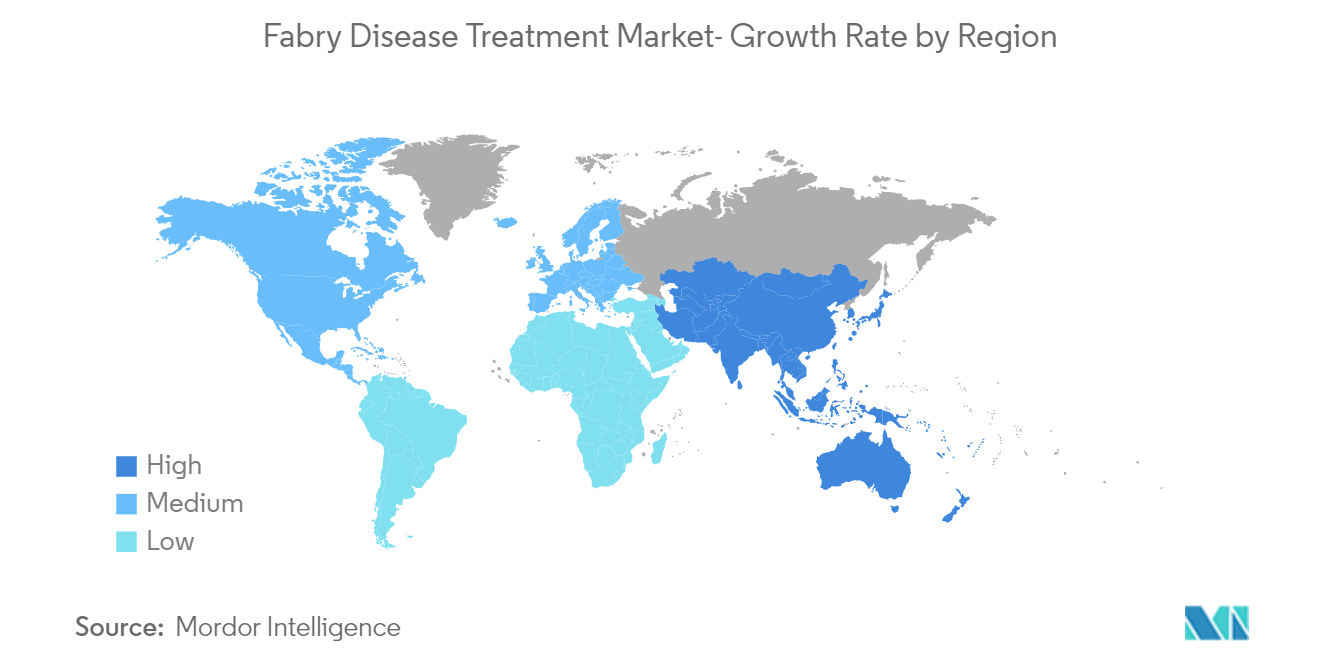
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
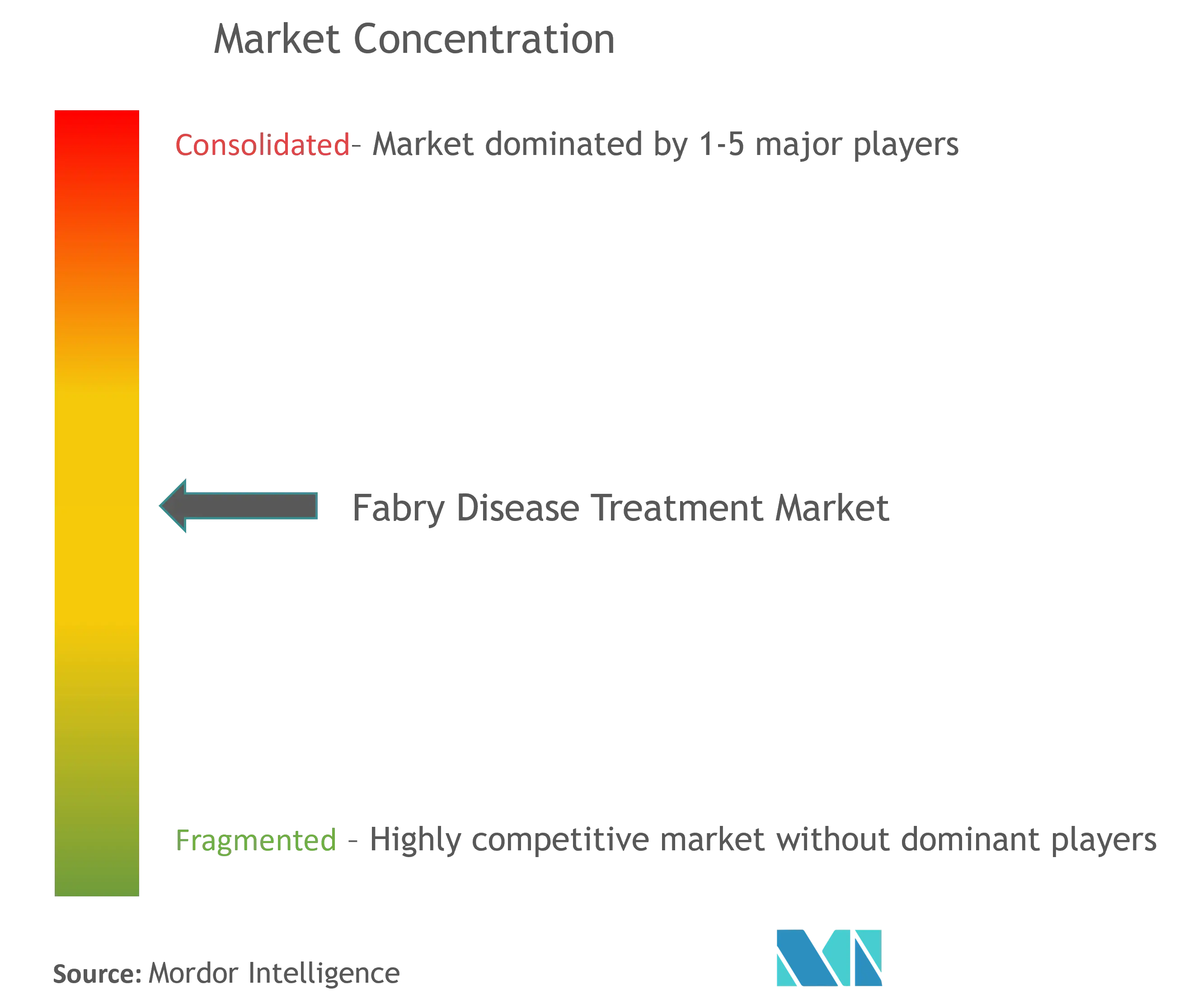
| Market Concentration | Low |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Fabry Disease Treatment Market Analysis
The Fabry disease treatment market is expected to register a CAGR of 6.8% over the forecast period.
COVID-19 significantly impacted the Fabry disease treatment market since Fabry disease patients receiving treatment in the hospitals experienced delayed drug infusion or disruptions in drug infusion services provided by the healthcare staff during the early pandemic period. Also, the COVID-19-infected Fabry disease patients witnessed changes in their immune system functions. For instance, according to the article published in June 2022 in PubMed, in Fabry Disease (FD) patients, the risk for severe COVID-19 was driven by the immune system rather than by FD itself. Thus, the complication of FD was exacerbated due to COVID-19; however, the unavailability of immediate medication significantly impacted the market growth. Besides, the market is expected to witness significant growth in the coming years owing to the rise in advancements in FD treatment and strong pipeline assets by the key market players.
The factor contributing to the market's growth includes a surge in awareness associated with Fabry disease treatment, a rise in R&D related to Fabry disease, and promising pipeline products, which drives the market's growth. Awareness associated with Fabry's disease is crucial for patients and healthcare staff. In recent years, there has been a rise in awareness related to treating Fabry disease in different countries such as the United States, United Kingdom, and others. For instance, according to the National Fabry Disease Foundation (NFDF), 'April' is observed as Fabry Disease Awareness Month annually in the United States. The awareness programs include educating the public, patients, caregivers, and healthcare professionals. Furthermore, the awareness activities help to enhance the perception, diagnosis, and management of Fabry disease, which is thereby expected to drive market growth over the study period.
In addition, the rise in research and development and increase in funding related to Fabry disease treatment is also expected to fuel the market growth. For instance, in December 2022, Bio Sidus SA sponsored a clinical trial to evaluate the efficacy and safety of AGA BETA BS in patients with FD already treated and previously stabilized with Fabrazyme. Also, various Fabry disease pipeline products that have the potential to receive regulatory approvals also drive the growth of the market. For instance, in August 2021, the European Commission approved Galafold, marketed by Amicus Therapeutics, for the long-term treatment of Fabry disease patients aged 12 years and above with an amenable mutation.
Thus, owing to the increased awareness of FD, the rise in product launches, and regulatory approval for FD therapies, the studied market is expected to witness significant growth over the study period. However, the lack of diagnostic resources in emerging countries will impede market growth over the study period.
Fabry Disease Treatment Market Trends
Enzyme Replacement Therapy is Expected to Witness a Healthy Growth Over the Forecast Period
Patients with Fabry disease may benefit clinically from enzyme replacement therapy when treatment is started early before the development of organ damage such as chronic kidney disease or cardiac fibrosis. The two significant enzyme replacement therapies employed in treating Fabry disease include Agalsidase Beta and Agalsidase Alfa. Furthermore, upon administration, both of these mimic the actions of alpha-galactosidase A.
The studied segment is expected to grow significantly due to the higher demand for enzyme replacement therapies in treating Fabry disease and their positive outcome in patients. This is because enzyme replacement therapies such as Fabrazyme work on the primary cause of Fabry disease. For instance, Fabrazyme addresses mutations in the GLA gene, which causes the lack of alpha-galactosidase. According to an article published in October 2022 in Frontier Journal, ERT was used successfully to treat Fabry disease and entails the exogenous replacement of the GLA enzyme. Furthermore, studies demonstrated the probability of a more extended life period when enzyme replacement therapies (ERTs) are employed for FD. For instance, as per the article published in May 2022 in IntechOpen journal, enzyme replacement therapy is possible to cure FD, giving patients substantially longer life expectancies and less severe systemic symptoms than in the past. Thus, owing to the increased adoption of enzyme replacement therapies in FD, the demand for ERT is likely to rise, contributing to segment growth over the forecast period.
In addition, pharmaceutical companies are adopting strategies to market new agalsidase beta drugs to treat Fabry disease. For instance, in March 2022, JCR Pharmaceuticals Co., Ltd. entered into an agreement with Sumitomo Dainippon Pharma Co., Ltd. for marketing Agalsidase Beta BS I.V. Infusion in Japan to treat Fabry disease. Thus, owing to such a strategic agreement by the key players, the availability of ERT is expected to rise, further boosting the market growth over the study period.
Hence, owing to the rise in advantages offered by the ERT in FD and various strategic agreements by the key players, the studied segment is expected to witness significant market growth over the study period.
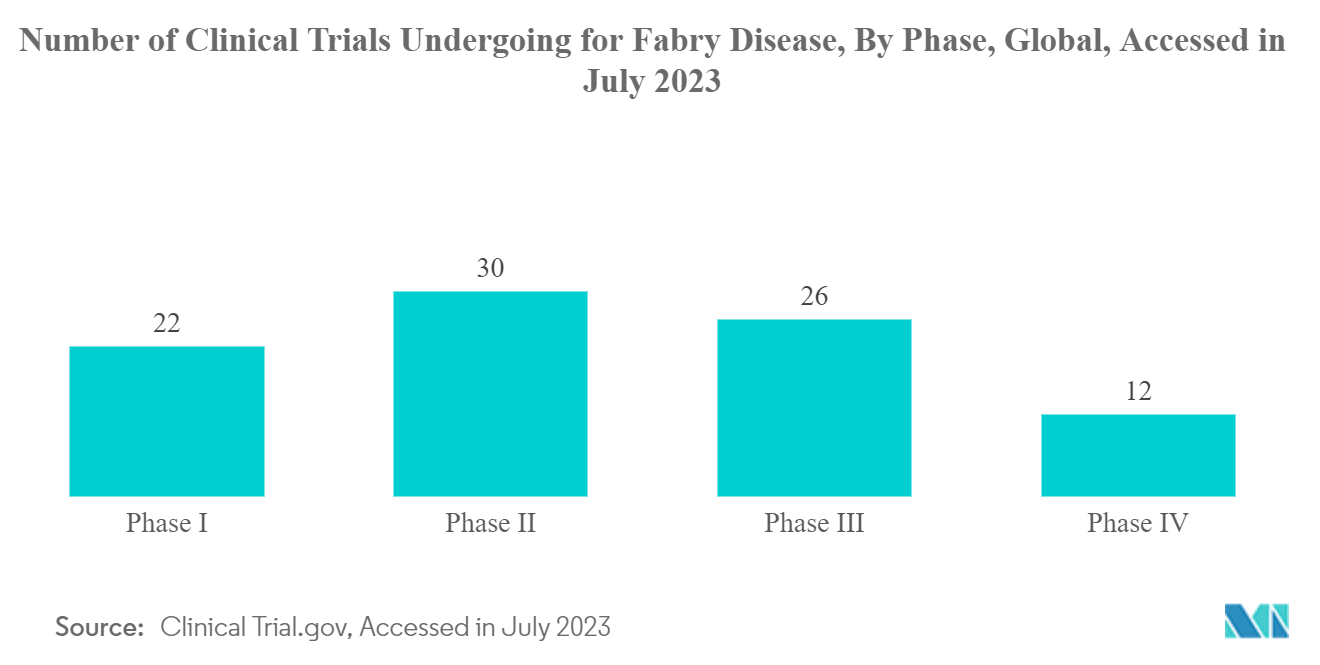
North America is Expected to Witness a Significant Growth Over the Forecast Period
North America is expected to witness healthy growth over the forecast period owing to the high healthcare expenditure, rise in awareness related to Fabry disease, and increase in pipeline assets and product launches of key market players in the region. For instance, in March 2022, Genzyme initiated a clinical trial to evaluate the effect of venglustat on neuropathic and abdominal pain symptoms of Fabry disease in adult participants with Fabry disease who are treatment-naïve or untreated for at least 6 months. Such clinical trials may lead to positive results leading to the introduction of new products in the market in the future and hence may contribute to the growth of the market in the future.
Furthermore, government initiatives to fund and support rare diseases across the region are expected to bolster market growth over the study period. For instance, in March 2023, the Minister of Health Canada publicized measures to support one of the first National Strategies for Drugs for Rare Diseases, with an investment of up to USD 1.5 billion over three years. Through this, the government increases access to and affordability of effective drugs for rare diseases to improve the health of patients across Canada, including children.
Therefore, owing to the rise in pipeline studies related to FD therapies and high investment from the government to support rare diseases, the studied market is expected to witness significant growth in North America over the forecast period.
Fabry Disease Treatment Industry Overview
The Fabry disease treatment market is consolidated in nature due to the presence of a few major players operating globally and regionally. A few of the major companies in the market are Sanofi (Genzyme Corporation), Takeda Pharmaceutical Company Limited, Amicus Therapeutics, Inc, ISU ABXIS, JCR Pharmaceuticals Co., Ltd., Protalix BioTherapeutics, among others.
Fabry Disease Treatment Market Leaders
-
Sanofi (Genzyme Corporation)
-
Takeda Pharmaceutical Company Limited
-
Amicus Therapeutics, Inc
-
ISU ABXIS
-
JCR Pharmaceuticals Co., Ltd.
*Disclaimer: Major Players sorted in no particular order
Fabry Disease Treatment Market News
- May 2023: Chiesi Global Rare Diseases and Protalix BioTherapeutics, Inc. received FDA approval for Elfabrio (pegunigalsidase alfa-iwxj) in the United States for the treatment of adult patients with Fabry disease. Elfabrio is supplied as a preservative-free solution in a single-dose vial. Each vial contains 20mg/10mL of pegunigalsidase alfa-iwxj. Treatment is administered by intravenous infusion every 2 weeks.
- May 2023: Sangamo Therapeutics, Inc., a genomic medicine company, received Fast Track Designation from the FDA for isaralgagene civaparvovec, or ST-920, a wholly owned gene therapy product candidate for the treatment of Fabry disease. ST-920 is currently being evaluated in the Phase 1/2 STAAR study, with a total of 20 patients dosed to date.
Fabry Disease Treatment Market Report - Table of Contents
1. INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
- 4.1 Market Overview
-
4.2 Market Drivers
- 4.2.1 Surge in Awareness Associated with Fabry Disease
- 4.2.2 Rise in R&D Related to Fabry Disease Coupled with Promising Pipeline Products
-
4.3 Market Restraints
- 4.3.1 Lack of Diagnostic Resources in Emerging Countries
-
4.4 Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
-
5.1 By Treatment
- 5.1.1 Enzyme Replacement Therapy
- 5.1.2 Oral Chaperone Therapy
- 5.1.3 Other Treatments
-
5.2 By Route of Aministration
- 5.2.1 Oral Route
- 5.2.2 Intravenous Route
-
5.3 By Distribution Channel
- 5.3.1 Hospital Pharmacies
- 5.3.2 Retail Pharmacies
- 5.3.3 Online Pharmacies
-
5.4 Geography
- 5.4.1 North America
- 5.4.1.1 United States
- 5.4.1.2 Canada
- 5.4.1.3 Mexico
- 5.4.2 Europe
- 5.4.2.1 Germany
- 5.4.2.2 United Kingdom
- 5.4.2.3 France
- 5.4.2.4 Italy
- 5.4.2.5 Spain
- 5.4.2.6 Rest of Europe
- 5.4.3 Asia-Pacific
- 5.4.3.1 China
- 5.4.3.2 Japan
- 5.4.3.3 India
- 5.4.3.4 Australia
- 5.4.3.5 South Korea
- 5.4.3.6 Rest of Asia-Pacific
- 5.4.4 Rest of the World
6. COMPETITIVE LANDSCAPE
-
6.1 Company Profiles
- 6.1.1 Sanofi (Genzyme Corporation)
- 6.1.2 Takeda Pharmaceutical Company Limited
- 6.1.3 Amicus Therapeutics, Inc
- 6.1.4 ISU ABXIS
- 6.1.5 JCR Pharmaceuticals Co., Ltd.
- 6.1.6 Protalix BioTherapeutics
- 6.1.7 Chiesi Farmaceutici S.p.A.
- 6.1.8 Freeline Therapeutics Holdings PLC
- 6.1.9 Yuhan Corporation
- 6.1.10 M6P Therapeutics
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
** Subject To AvailablityFabry Disease Treatment Industry Segmentation
As per the scope of the report, Fabry disease is defined as a rare genetic condition that is marked by the deficiency of an enzyme called alpha-galactosidase A. The lower levels or absence of alpha-galactosidase A causes the accumulation of globotriaosylceramide (GL-3) in the affected tissues of the central nervous system, heart, kidneys, and skin. The Fabry disease treatment market is segmented by treatment (enzyme replacement therapy, oral chaperone therapy, and other treatments), route of administration (oral route and intravenous route), distribution channel (hospital pharmacies, retail pharmacies, and online pharmacies), and geography (North America, Europe, Asia-Pacific and Rest of the World). The report offers the value (in USD) for the above segments.
| By Treatment | Enzyme Replacement Therapy | |
| Oral Chaperone Therapy | ||
| Other Treatments | ||
| By Route of Aministration | Oral Route | |
| Intravenous Route | ||
| By Distribution Channel | Hospital Pharmacies | |
| Retail Pharmacies | ||
| Online Pharmacies | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Geography | Europe | Germany |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Geography | Asia-Pacific | China |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Geography | Rest of the World |
Fabry Disease Treatment Market Research FAQs
What is the current Global Fabry Disease Treatment Market size?
The Global Fabry Disease Treatment Market is projected to register a CAGR of 6.80% during the forecast period (2024-2029)
Who are the key players in Global Fabry Disease Treatment Market?
Sanofi (Genzyme Corporation), Takeda Pharmaceutical Company Limited, Amicus Therapeutics, Inc, ISU ABXIS and JCR Pharmaceuticals Co., Ltd. are the major companies operating in the Global Fabry Disease Treatment Market.
Which is the fastest growing region in Global Fabry Disease Treatment Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Global Fabry Disease Treatment Market?
In 2024, the North America accounts for the largest market share in Global Fabry Disease Treatment Market.
What years does this Global Fabry Disease Treatment Market cover?
The report covers the Global Fabry Disease Treatment Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Global Fabry Disease Treatment Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Global Fabry Disease Treatment Industry Report
Statistics for the 2024 Fabry Disease Treatment market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Fabry Disease Treatment analysis includes a market forecast outlook to for 2024 to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.



