Market Trends of Global Intravenous Immunoglobulin Industry
This section covers the major market trends shaping the Intravenous Immunoglobulin Market according to our research experts:
The Primary Immunodeficiency Diseases Segment is Expected to Register a High CAGR Over the Forecast Period
By application, the primary immunodeficiency disease (PIDs) segment is expected to hold a significant market share.
Intravenous immunoglobulin (IVIG) therapy has been majorly used in treating primary immunodeficiency diseases (PIDs) and other immune deficiency disorders over the past few years. There are several specific medical therapies available for people with PIDs with antibody deficiency, which account for more than half of the primary immunodeficiencies. These illnesses, such as common variable immune deficiency (CVID), X-linked agammaglobulinemia (XLA), and other disorders, are characterized by a lack of and/or impaired antibody function. The high burden of primary immunodeficiency is a major factor fueling the segment's growth. For instance, as per the research article titled "Systematic Review of Primary Immunodeficiency Diseases in Malaysia: 1979-2020", published in Frontiers in Immunology in August 2020, approximately six million people are predicted to be living with PIDs globally, among which only 27,000-60,000 cases have been diagnosed. As per the above-mentioned source, the incidence of PIDs in Singapore was observed to be 2.65 per 100,000 live births, with an estimated occurrence rate of one in 37,000 live births. Also, as per an April 2020 update by the Centers for Disease Control and Prevention (CDC), there are more than 400 types of primary immunodeficiencies that vary in severity. Thus, these statistics reflect the rising need for immunoglobulins for the treatment of immunodeficiency disorders. Hence, the growing burden of PIDs globally is expected to drive the studied segment's growth over the forecast period.
Furthermore, several pharmaceutical companies are engaged in developing immunoglobulin products for the treatment of primary immunodeficiency. For instance, in May 2021, GC Pharma reported that the United States Food and Drug Administration (FDA) accepted the company's Biologics License Application (BLA) for "GC5107 (Immune Globulin Intravenous (Human), 10% Liquid)" intended for the treatment of Primary Humoral Immunodeficiency, a class of inherited genetic disorders that causes an individual to have a deficient or absent immune system.
Hence, with the availability of IVIG therapies and the advancements in intravenous immunoglobulin products to treat the highly prevalent primary immunodeficiency diseases, the studied segment is expected to grow significantly over the coming years.
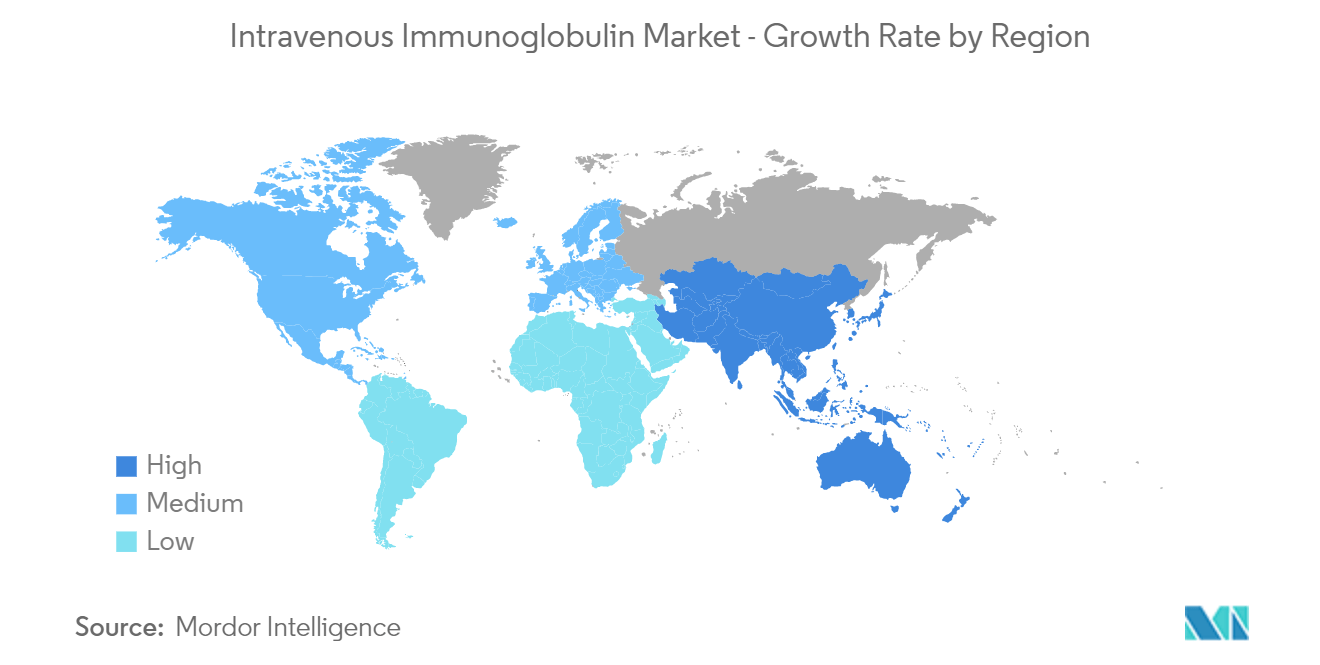
North America is Expected to Dominate the Market Over The Forecast Period
The United States is expected to hold a major share of the market in North America. The major factors propelling the market's growth in the country are a developed and well-structured healthcare system; a growing focus on research and development; and the strong presence of market players.
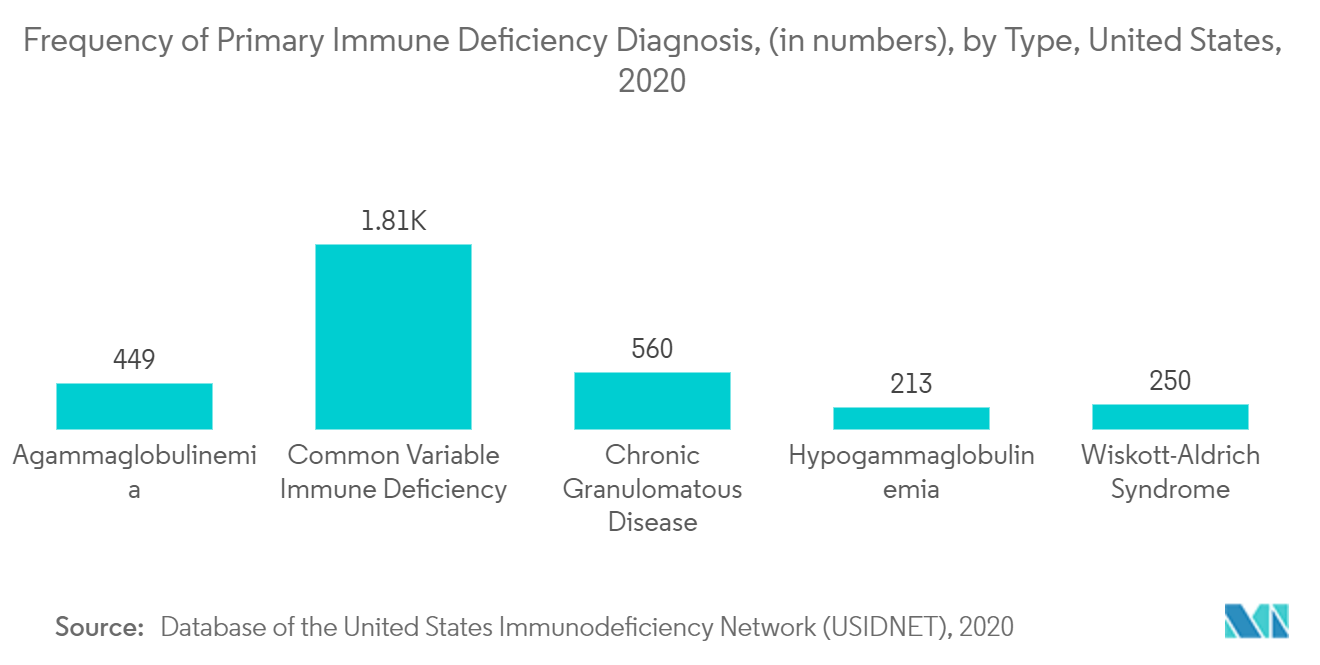
Additionally, the growing burden of target diseases such as primary immunodeficiency diseases in the region is expected to boost the market's growth. For instance, according to the National Institute of Allergy and Infectious Diseases, in 2020, more than 400 types of varied PIDs were diagnosed in the United States, accounting for more than 500,000 American populations affected by PIDs. Additionally, as per the USIDNET Registry, 2020, the frequency of severe combined immunodeficiency (SCID) in the American population was 355. Further, the increasing research and development activities and the rapid product approvals are predicted to drive the intravenous immunoglobulin market growth in the region.
Furthermore, in August 2021, Emergent BioSolutions Inc., a multinational specialty biopharmaceutical company based in the United States, reported the start of Phase III clinical trial to evaluate its investigational SARS-CoV-2 Immune Globulin Intravenous (COVID-HIG) plasma-derived therapy as a potential outpatient treatment for patients with coronavirus (COVID-19) who are at high risk of progression to severe disease.
Additionally, in February 2021, Pfizer received United States Food and Drug Administration approval for the supplemental Biologics License Application (sBLA), which was submitted for PANZYGA, an intravenous immunoglobulin (IVIg) to treat adult patients with chronic inflammatory demyelinating polyneuropathy (CIDP).
Thus, the market is expected to witness significant growth during the forecast period due to the abovementioned factors.