Market Trends of Non-Invasive Prenatal Testing Industry
Down Syndrome Segment Dominates the Non-invasive Prenatal Testing Market
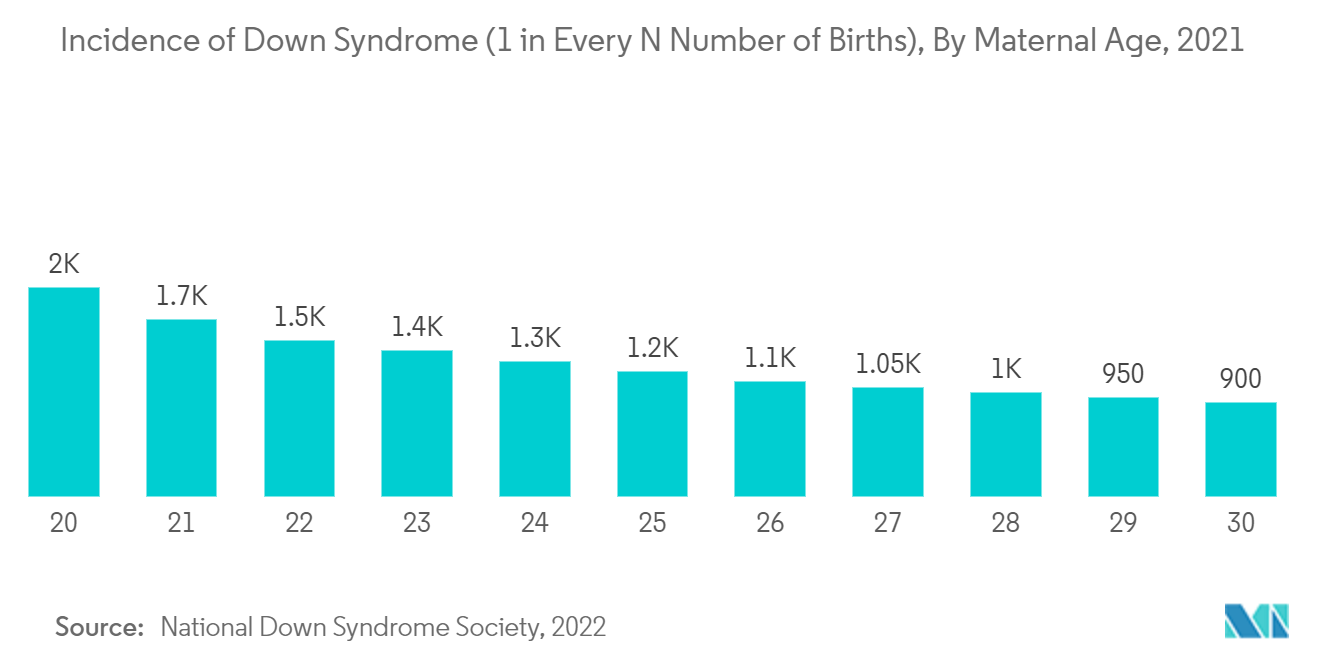
Down syndrome is a chromosomal disorder, also called trisomy 21, trisomy G, 47, XX,+21, and 47, XY,+21. It includes certain birth defects, learning problems, and deformities of facial features. A child with Down syndrome may also have heart defects and problems with vision and hearing. According to the Centers for Disease Control and Prevention data updated in April 2021, Down syndrome is the most common chromosomal condition diagnosed in the United States. Every year, about 6,000 babies born in the United States have Down syndrome. This means that Down syndrome occurs in about 1 out of every 700 babies. Hence, the growing incidence rate of Down syndrome is expected to create lucrative opportunities for the growth of the market studied during the forecast period.
Non-invasive prenatal testing consists of analyzing cell-free DNA (cfDNA) circulating in the mother's blood to detect Down syndrome. With the successful introduction of NIPT for detecting Down syndrome into routine prenatal care, it is important to understand the risks, benefits, and limitations to guide patients in making informed decisions. Non-invasive prenatal testing offers several clinical advantages over existing prenatal tests, such as maternal serum screening (MSS). According to the study by Stanislav Birko et al. published in January 2022, for the screening of Down syndrome, non-invasive prenatal testing (NIPT) can be performed as early as the ninth week of pregnancy, holds no risk of miscarriage, and can detect the presence of Down syndrome (trisomy 21) with high sensitivity (99.9%) and specificity (98%). As a result, the demand for these technologies has increased and is being adopted by many developing countries, which is expected to drive the growth of the segment.
Thus, the abovementioned factors are expected to increase market growth during the forecast period.
North America is Expected to Dominate the Market Over the Forecast Period
North America is expected to hold a leading position in the non-invasive prenatal testing (NIPT) market during the forecast period. High preterm birth rate, high maternal mortality rate, and rising incidences of chromosomal abnormalities will likely increase the adoption and demand for tests in this region. Various research studies provide insight into the effectiveness of awareness about prenatal care. For instance, in August 2022, a research study published in the National Library of Medicine stated that advances in antenatal care have resulted in increased awareness of fetal malformations, and malpresentation is expected to improve preparation for a high-risk delivery. Hence pregnant women need to adhere to prenatal care recommendations for optimal outcomes. Thus, North America is expected to dominate the non-invasive prenatal testing (NIPT) market over the forecast period due to the growing awareness of chromosomal abnormalities among fetuses.
Furthermore, high diagnosis rates, wider adoption and awareness among the people, and the introduction of new and technologically advanced tests are also contributing to the market growth in North America. For instance, in May 2021, Yourgene launched IONA Care, a non-invasive prenatal test (NIPT) service offering to measure whether a pregnant woman is carrying a fetus with sex chromosome aneuploidies (SCA) and autosomal aneuploidies (AA), in addition to the current IONA test, which offers screening for trisomies 21, 18, and 13 and fetal sex determination. Thus, such product launches are expected to increase market growth over the forecast period.
Hence, the abovementioned factor will have a positive influence on the growth of the market studied during the forecasted period.