Market Trends of North America Single-Use Medical Device Reprocessing Industry
This section covers the major market trends shaping the North America Single-Use Medical Device Reprocessing Market according to our research experts:
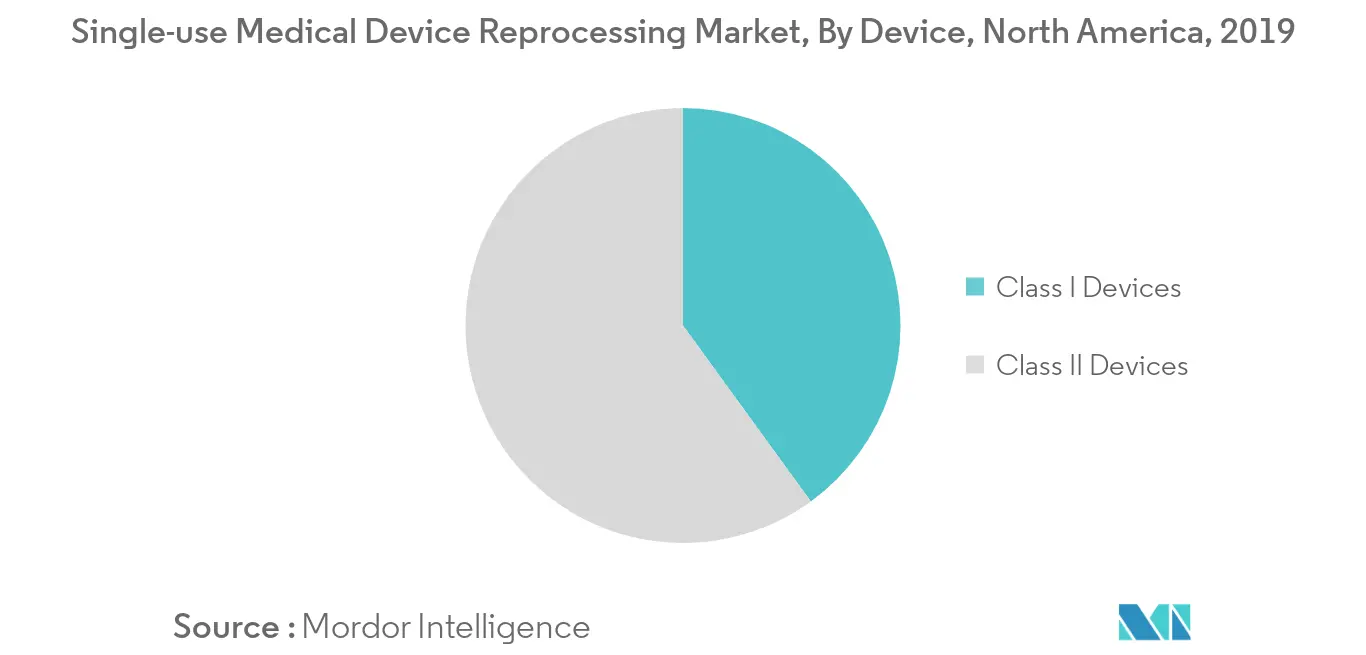
Sequential Compression Sleeves by Class II Device Segment is Poised to Register Robust Growth
- Sequential compression sleeves are designed to increase venous blood flow in patients with deep vein thrombosis and pulmonary embolism. Compression sleeves are considered reusable, as they can withstand the necessary cleaning and sterilization process.
- The quality of the device and physical characteristics are not likely to be affected by reprocessing and the device remains safe and effective for its intended use. Hospitals are believed to save USD 300,000 by reprocessing compression sleeves. FDA reported that in 25% of all hospitals in the United States that use SUDs, sequential compression devices constitute about 15.8% of the reprocessed items. Compression sleeves account for the greatest volume of the overall business of SUDs reprocessing.
- Stryker is the leading provider of reprocessed compression sleeve devices and has FDA clearance for the product offering. The company's reprocessed compression devices are intended for use in preventing deep vein thrombosis, reducing post-operative pain and swelling, enhancing blood circulation, and reducing wound healing time.
- The annual savings estimate of Stryker for DVT compressor sleeves is USD 27,950. Currie Medical Specialties Inc. and Covidien (Medtronic) have also received 510K FDA clearances to reprocess compression sleeves.
- Reprocessed Kendall SCD express compression sleeves and reprocessed Kendall SCD comfort compression sleeves from Covidien (Medtronic) received FDA clearance on January 19, 2017. Medline also offers reprocessed compression sleeves.