North America Uterine Cancer Diagnostics & Treatment Market Size
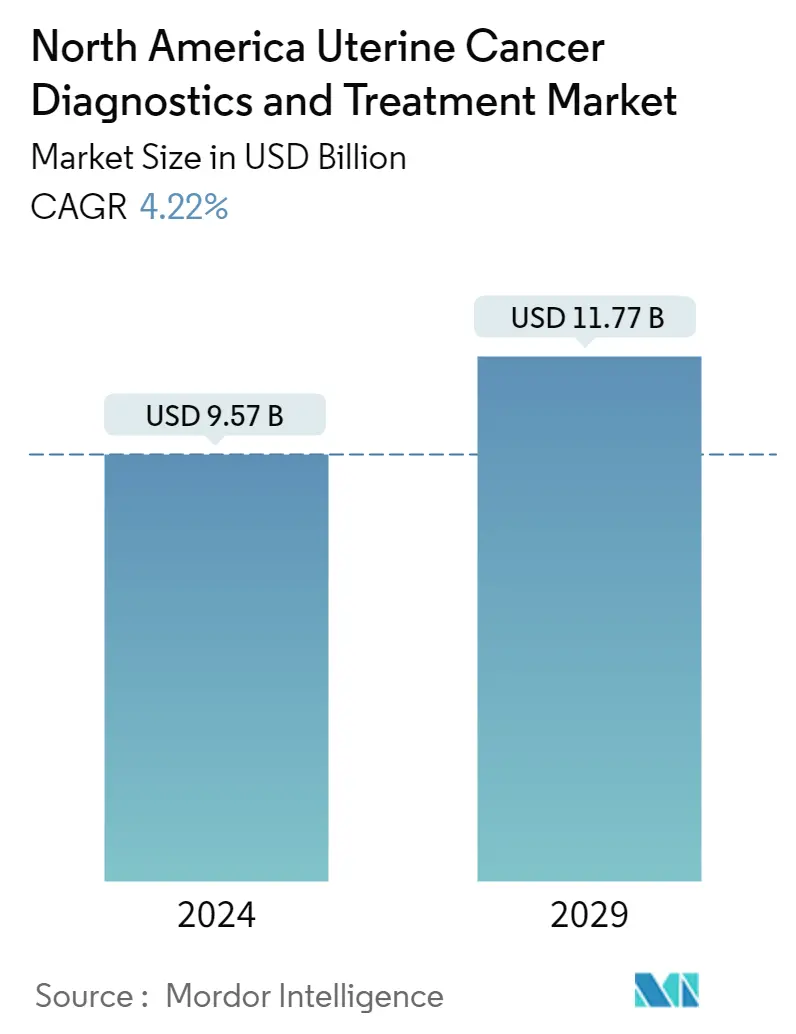
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Market Size (2024) | USD 9.57 Billion |
| Market Size (2029) | USD 11.77 Billion |
| CAGR (2024 - 2029) | 4.22 % |
| Market Concentration | Low |
Major Players
*Disclaimer: Major Players sorted in no particular order |
North America Uterine Cancer Diagnostics & Treatment Market Analysis
The North America Uterine Cancer Diagnostics & Treatment Market size is estimated at USD 9.57 billion in 2024, and is expected to reach USD 11.77 billion by 2029, growing at a CAGR of 4.22% during the forecast period (2024-2029).
The COVID-19 pandemic impacted the healthcare systems globally and significantly impacted the North American uterine cancer diagnostics and treatment market. For instance, according to the article published in November 2021 by Cancer Connect, doctors from Dana Farber Cancer Institute determined that during the COVID-19 pandemic, there was a 46.0% decrease in the diagnoses of the six most common cancer types - breast, colorectal, lung, pancreatic, gastric, and esophageal cancers in North America. Moreover, in the COVID-19 era, nearly 88.0% of the cancer care centers faced challenges in delivering usual cancer care for many reasons, including preventive measures, lack of personal protective equipment, and staff shortage, as per the NCBI research article published in 2021. However, with the resumption of all non-essential surgeries and medical requirements, the market has started to gain traction. It is expected to continue the upward trend over the forecast period.
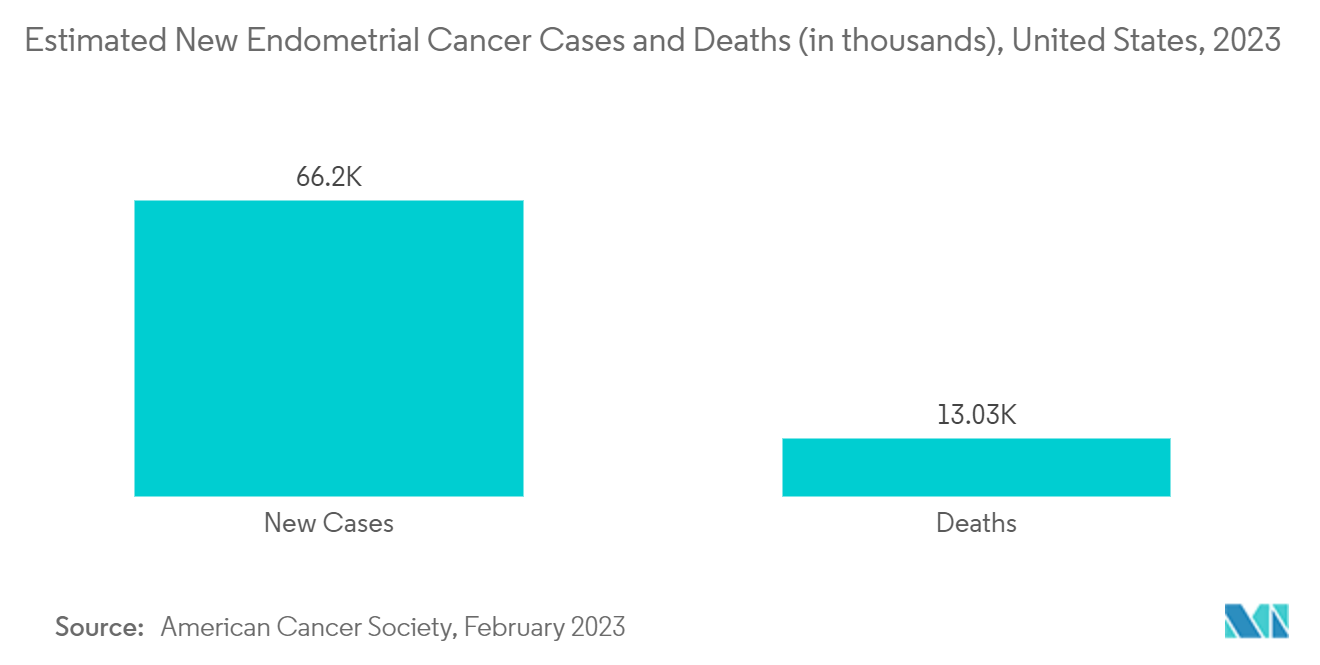
The major drivers for the market are the rising awareness about uterine cancer and the available therapies, increasing cases of uterine cancer, innovation in drug development, and subsequent technological advancements. For instance, according to the American Cancer Society estimates for cancer of the uterus in the United States for 2023, around 66,200 new cases of cancer of the body of the uterus (uterine body or corpus) will be diagnosed, and around 13,030 women will die from cancers of the uterine body in 2023. The same source also stated that endometrial cancer affects mainly post-menopausal women. The average age of women diagnosed with endometrial cancer is 60. It's uncommon in women under the age of 45. Thus, increasing cases of uterine cancer are expected to propel the demand for uterine cancer diagnostics and treatment, thereby propelling market growth.
The increasing product approval for treating uterine cancer diagnostics is also expected to propel the market growth over the forecast period. For instance, in April 2021, the U.S. FDA granted accelerated approval to Jemperli (dostarlimab) for treating patients with recurrent or advanced endometrial cancer that has progressed on or following prior treatment with platinum-containing chemotherapy and whose cancers have a specific genetic feature known as dMMR, as determined by an FDA-approved test. Similarly, Merck announced that the U.S. Food and Drug Administration (FDA) has approved KEYTRUDA, Merck’s anti-PD-1 therapy, as a single agent for the treatment of patients with advanced endometrial carcinoma that is microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), as determined by an FDA-approved test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. Thus, increasing product approvals is anticipated to positively impact the market growth over the forecast period.
However, the low success rate in clinical trials for cancer drugs, the high cost of research and development, and the high cost associated with the treatment will likely hinder the market growth over the forecast period.
North America Uterine Cancer Diagnostics & Treatment Market Trends
Immunotherapy Segment is Expected to Register Considerable Growth Over the Forecast Period
Immunotherapy is a treatment in which drugs help an individual’s immune system recognize and kill the cancer cells. Immunotherapy is done for treating certain forms of endometrial cancer that have spread or recurred. Immunotherapy is a treatment that uses a person's immune system to fight cancer. Immunotherapy can boost or change how the immune system works to find and attack cancer cells. Immunotherapy stimulates the endogenous immune response, specifically against tumor cells, and is the new frontier of anticancer treatment. Several compounds targeting different biological pathways are available. Some of these agents are already approved for treating non-gynecological malignancies like lung cancer and melanoma. They could also play a major role in the treatment of endometrial cancer.
Increasing product approval and rising product launches are the major drivers for segment growth. For instance, in March 2022, Karyopharm Therapeutics Inc., a commercial-stage pharmaceutical company pioneering novel cancer therapies, provided an update on its discussions with the U.S. Food and Drug Administration (FDA) regarding its previously planned supplemental New Drug Application (sNDA) submission based on the data from the Phase 3 SIENDO study evaluating selinexor as a front-line maintenance therapy following chemotherapy in patients with advanced or recurrent endometrial cancer. Similarly, in March 2022, the Food and Drug Administration approved pembrolizumab (Keytruda, Merck) as a single agent for patients with advanced endometrial carcinoma that is microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), as determined by an FDA-approved test, who have disease progression following prior systemic therapy in any setting and who are not candidates for curative surgery or radiation. Thus, owing to such instances, the segment is anticipated to have considerable segment growth over the forecast period.
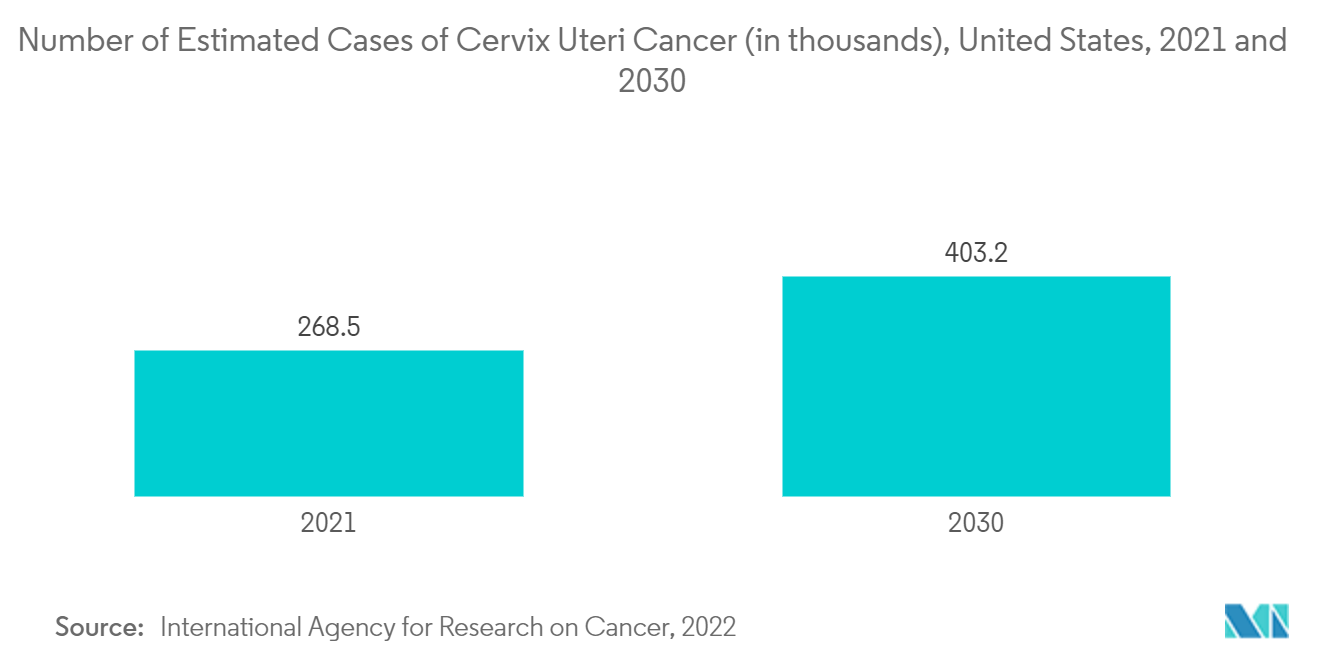
United States is Expected to Witness Considerable Growth Over the Forecast Period
Key product launches, high concentration of market players or manufacturer's presence, acquisition & partnerships among major players, and increasing healthcare expenditure in the United States are some factors driving the growth of the United States uterine cancer diagnostics and treatment market. For instance, as per the United States Research and Development Funding and Performance: Fact Sheet, 2022, the R&D expenditures in the United States in 2021 were estimated at USD 580 billion, of which USD 96.5 billion was invested in basic research, USD 115.0 billion on applied research, and USD 368.5 billion was employed in the development sector. Such high investment is expected to accelerate the research, development, and approval of uterine cancer drugs in the country, propelling market growth.
The increasing product approvals and launches in the country are also anticipated to increase market growth. For instance, in August 2022, Myovant Sciences and Pfizer received FDA approval in the United States for MYFEMBREE, a once-daily treatment for managing moderate to severe pain associated with endometriosis. MYFEMBREE is also approved for heavy menstrual bleeding associated with uterine fibroids in premenopausal women. Myovant and Pfizer will continue co-commercializing MYFEMBREE in the United States, and the product will be available immediately. Thus, owing to such factors, the country's market is expected to grow considerably.
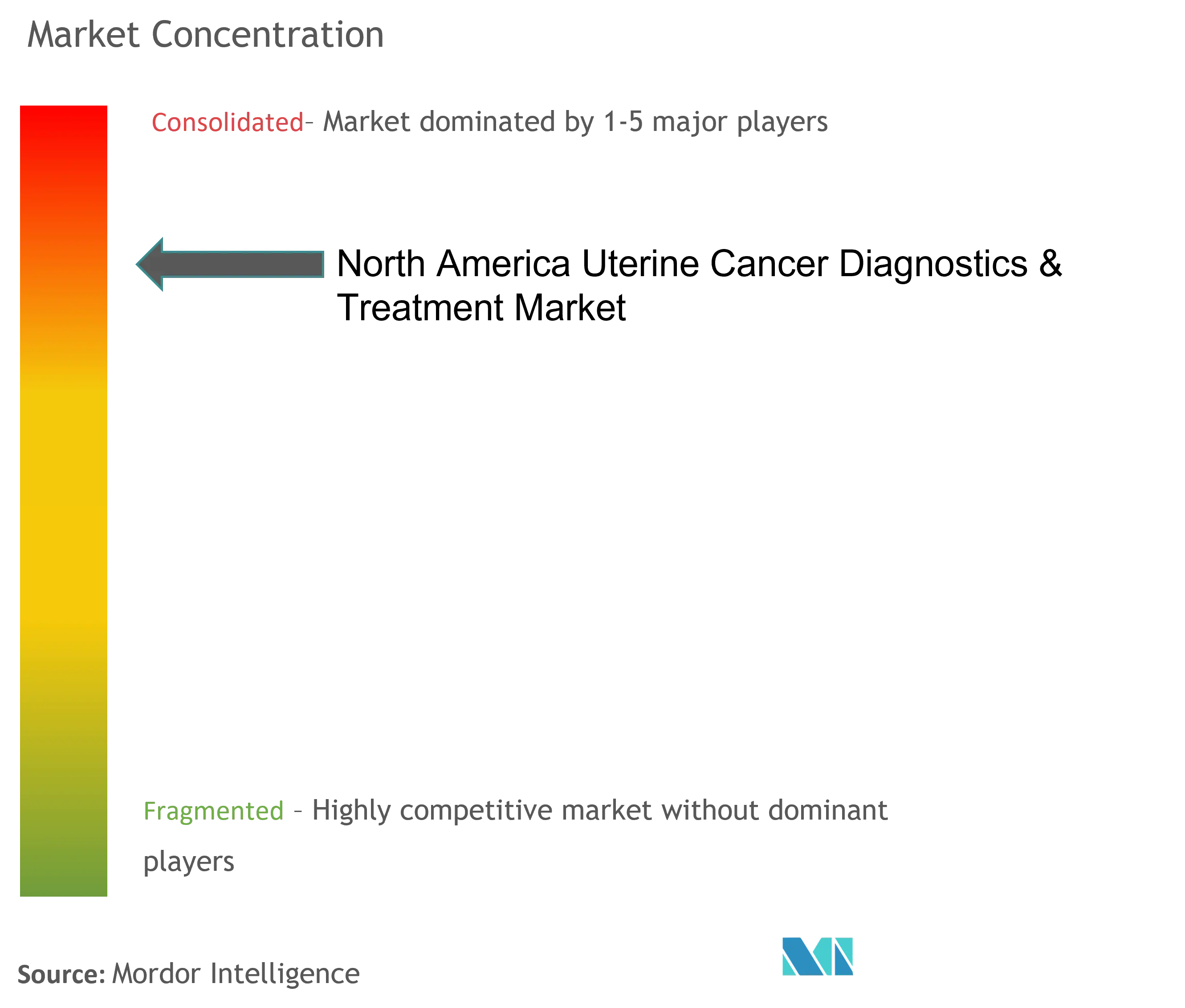
North America Uterine Cancer Diagnostics & Treatment Industry Overview
The uterine cancer diagnostics and treatment market comprises global players that focus on the oncology division and are consolidated. Some major companies in this market include Merck & Co. Ltd, Pfizer Inc., and AbbVie Inc., among others. With the rising research by the pharmaceutical industry on the development of better therapeutics and diagnostics for various cancers, more companies are believed to enter the market in the coming future.
North America Uterine Cancer Diagnostics & Treatment Market Leaders
-
AbbVie Inc.
-
Merck & Co. Ltd
-
Pfizer Inc.
-
Becton Dickinson and Company
-
Bristol-Myers Squibb Company
*Disclaimer: Major Players sorted in no particular order
North America Uterine Cancer Diagnostics & Treatment Market News
- February 2023: GSK announced that the United States Food and Drug Administration (FDA) has granted full approval for Jemperli (dostarlimab-gxly) for the treatment of adult patients with recurrent or advanced mismatch repair-deficient endometrial cancer (dMMR) as determined by the United States FDA.
- March 2022: The Food and Drug Administration has approved pembrolizumab (Keytruda, Merck) as monotherapy for patients with advanced endometrial cancer that is microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), as determined by an FDA-approved test, who have progression disease after prior systemic therapy in any setting and are not candidates for curative surgery or radiation.
North America Uterine Cancer Diagnostics & Treatment Market Report - Table of Contents
1. INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
- 4.1 Market Overview
-
4.2 Market Drivers
- 4.2.1 Rising Awareness about Uterine Diseases and the Available Therapies
- 4.2.2 Increasing Health Care Expenditure
- 4.2.3 Innovation in Drug Development and Subsequent Technological Advancements
-
4.3 Market Restraints
- 4.3.1 Low Success Rate in Clinical Trials for Cancer Drugs and High Cost of Research and Development
- 4.3.2 High Cost Associated with the Treatment
-
4.4 Porter's Five Forces Analysis
- 4.4.1 Bargaining Power of Buyers/Consumers
- 4.4.2 Bargaining Power of Suppliers
- 4.4.3 Threat of New Entrants
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value – USD)
-
5.1 By Cancer Type
- 5.1.1 Endometrial Cancer
- 5.1.2 Uterine Sarcoma
-
5.2 By Procedure
- 5.2.1 Treatment
- 5.2.1.1 Surgery
- 5.2.1.2 Immunotherapy
- 5.2.1.3 Radiation Therapy
- 5.2.1.4 Chemotherapy
- 5.2.1.5 Other Treatments
- 5.2.2 Diagnostics
- 5.2.2.1 Biopsy
- 5.2.2.2 Ultrasound
- 5.2.2.3 Hysteroscopy
- 5.2.2.4 Dilation and Curettage
- 5.2.2.5 Other Diagnostics
-
5.3 Geography
- 5.3.1 North America
- 5.3.1.1 United States
- 5.3.1.2 Canada
- 5.3.1.3 Mexico
6. COMPETITIVE LANDSCAPE
-
6.1 Company Profiles
- 6.1.1 AbbVie Inc.
- 6.1.2 Becton, Dickinson and Company
- 6.1.3 Bristol-Myers Squibb Company
- 6.1.4 F. Hoffmann-La Roche Ltd
- 6.1.5 GlaxoSmithKline PLC
- 6.1.6 Merck & Co. Inc.
- 6.1.7 Novartis AG
- 6.1.8 Pfizer Inc.
- 6.1.9 Takeda Pharmaceutical Company Limited
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
** Subject To AvailablityNorth America Uterine Cancer Diagnostics & Treatment Industry Segmentation
As per the scope of the report, uterine cancer is a common form of cancer of the female reproductive system that initiates in the lining of the uterus. Risk factors associated with this cancer are being overweight and starting periods at an early age.
North America's uterine cancer diagnostics and treatment market is segmented by cancer type (endometrial cancer, uterine sarcoma), procedure (surgery, immunotherapy, radiation therapy, chemotherapy, and other treatments), diagnostics (biopsy, ultrasound, hysteroscopy, dilation and curettage, and other diagnostics), and geography (North America, Europe, Asia-Pacific, the Middle East, Africa, and South America).
The report offers the value (in USD) for the above segments.
| By Cancer Type | Endometrial Cancer | |
| Uterine Sarcoma | ||
| By Procedure | Treatment | Surgery |
| Immunotherapy | ||
| Radiation Therapy | ||
| Chemotherapy | ||
| Other Treatments | ||
| By Procedure | Diagnostics | Biopsy |
| Ultrasound | ||
| Hysteroscopy | ||
| Dilation and Curettage | ||
| Other Diagnostics | ||
| Geography | North America | United States |
| Canada | ||
| Mexico |
North America Uterine Cancer Diagnostics & Treatment Market Research FAQs
How big is the North America Uterine Cancer Diagnostics & Treatment Market?
The North America Uterine Cancer Diagnostics & Treatment Market size is expected to reach USD 9.57 billion in 2024 and grow at a CAGR of 4.22% to reach USD 11.77 billion by 2029.
What is the current North America Uterine Cancer Diagnostics & Treatment Market size?
In 2024, the North America Uterine Cancer Diagnostics & Treatment Market size is expected to reach USD 9.57 billion.
Who are the key players in North America Uterine Cancer Diagnostics & Treatment Market?
AbbVie Inc., Merck & Co. Ltd, Pfizer Inc., Becton Dickinson and Company and Bristol-Myers Squibb Company are the major companies operating in the North America Uterine Cancer Diagnostics & Treatment Market.
What years does this North America Uterine Cancer Diagnostics & Treatment Market cover, and what was the market size in 2023?
In 2023, the North America Uterine Cancer Diagnostics & Treatment Market size was estimated at USD 9.18 billion. The report covers the North America Uterine Cancer Diagnostics & Treatment Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the North America Uterine Cancer Diagnostics & Treatment Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
North America Uterine Cancer Diagnostics & Treatment Industry Report
Statistics for the 2024 North America Uterine Cancer Diagnostics & Treatment market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. North America Uterine Cancer Diagnostics & Treatment analysis includes a market forecast outlook 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.



