Market Trends of Global Orphan Drugs Industry
This section covers the major market trends shaping the Orphan Drugs Market according to our research experts:
Neurology Segment is Anticipated to Witness Growth Over the Forecast Period
Neurological disorders make up nearly half of these rare diseases. Due to the lack of screening tests, diagnostic genomic sequencing, and specialized clinical expertise, rare central nervous system (CNS) disorders are frequently challenging to identify and treat. There are about 5,000-8,000 rare diseases. Some neurological disorders include reflex sympathetic dystrophy syndrome, Battaglia-Neri syndrome, Creutzfeldt Jakob Disease, agnosia, Aicardi syndrome, and Aicardi-Goutieres syndrome. As per the report titled "Prevalence and incidence of rare diseases: Bibliographic data", published in January 2022, the prevalence of chronic inflammatory demyelinating polyneuropathy is 3.7 in Europe. However, the diagnosis of rare neurological diseases involves several barricades, such as a lack of awareness about rare neurological diseases within the population. Rare diseases are creating huge healthcare and economic burden worldwide.
The National Organization for Rare Disorders (NORD) data updated in June 2020 estimates that there are more than 7,000 rare diseases that are affecting more than 30 million Americans. In addition, about one-third of these are thought to include neurological components and symptoms. Additionally, as per the data published by the Center for Rare Neurological Disease Research in January 2021, rare neurological diseases affect approximately 200,000 individuals in the United States. Thus, the growing burden of rare neurological diseases drives the demand for orphan drugs.
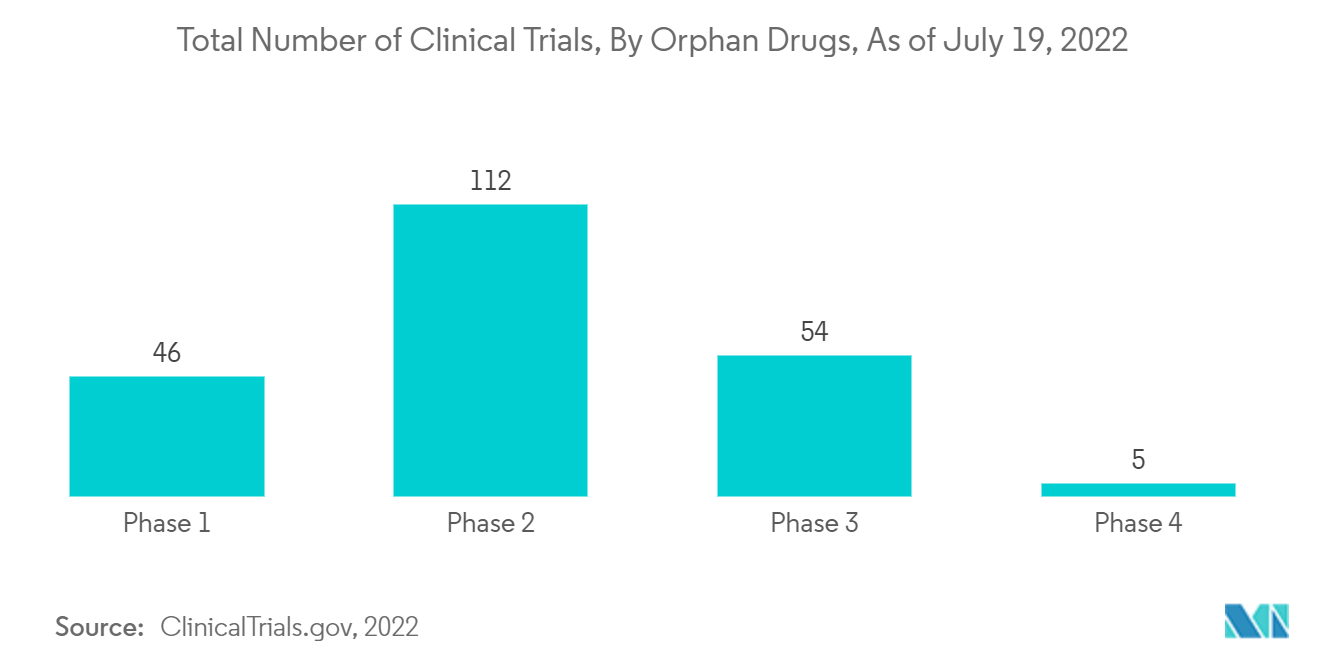
According to the study titled "Using four decades of FDA orphan drug designations to describe trends in rare disease drug development: substantial growth seen in the development of drugs for rare oncologic, neurologic, and pediatric-onset diseases" published in the Orphanet of the Rare Diseases in July 2021, oncology (1910, 37%), neurology (674, 13%), and infectious diseases (436, 9%) were the top three therapeutic categories represented in the orphan drug designations.
Moreover, the growing approval of the Food and Drug Administration (FDA) for the orphan drug is anticipated to boost the segment growth. For Instance, in November 2021, PharmaTher Holdings Ltd. to receive Orphan Drug Designation ('ODD') for ketamine to treat Status Epilepticus, a rare neurological disorder requiring emergency treatment for a seizure.
Thus, all aforementioned factors are expected to boost the segment growth over the forecast period.
North America Dominates the Market and Expected to do the same in the Forecast Period
North America currently dominates the market for orphan drugs and is expected to continue its stronghold for a few more years. In the North American region, the United States holds the largest market share. One of the reasons for market growth is that in the United States, a drug-designated orphan drug status receives seven years of marketing exclusivity upon FDA approval for a specific indication, tax credits, and user fee waiver.
Furthermore, rare diseases are found in around 30 million Americans, according to data updated in May 2022 by the Genetic and Rare Diseases Information Center (GARD), and 3.1 million Canadians, according to the Canadian Organization for Rare Disorders (CORD) 2021 update. Due to the high prevalence in America, the government has provided support for patients with rare diseases through research grants and funds. With only 5% of rare diseases having FDA-approved treatment, the NIH has provided USD 31 million in grants to study rare diseases between 2019 and 2020. Similarly, in October 2020, the Food and Drug Administration (FDA) carried out the Orphan Drug Act and awarded over USD 16 million for six clinical trial research studies to industry and academia for the next four years.
Moreover, a growing number of drugs with the orphan drug designation is anticipated to boost the market growth. For instance, in June 2020, Agios Pharmaceuticals Inc., received the United States Food and Drug Administration (FDA) orphan drug designation for their pipeline candidate of Mitapivat for the treatment of Thalassemia. In May 2020, AstraZeneca Plc and Daiichi Sankyo Company received United States Food and Drug Administration (FDA) orphan drug designation for Enhertu (Trastuzumab deruxtecan) for the treatment of patients with gastric cancer, including gastroesophageal junction cancer.
Therefore, owing to the factors mentioned above the orphan drugs market is anticipated to grow in the North American region.