Pediatric Vaccines Market Size
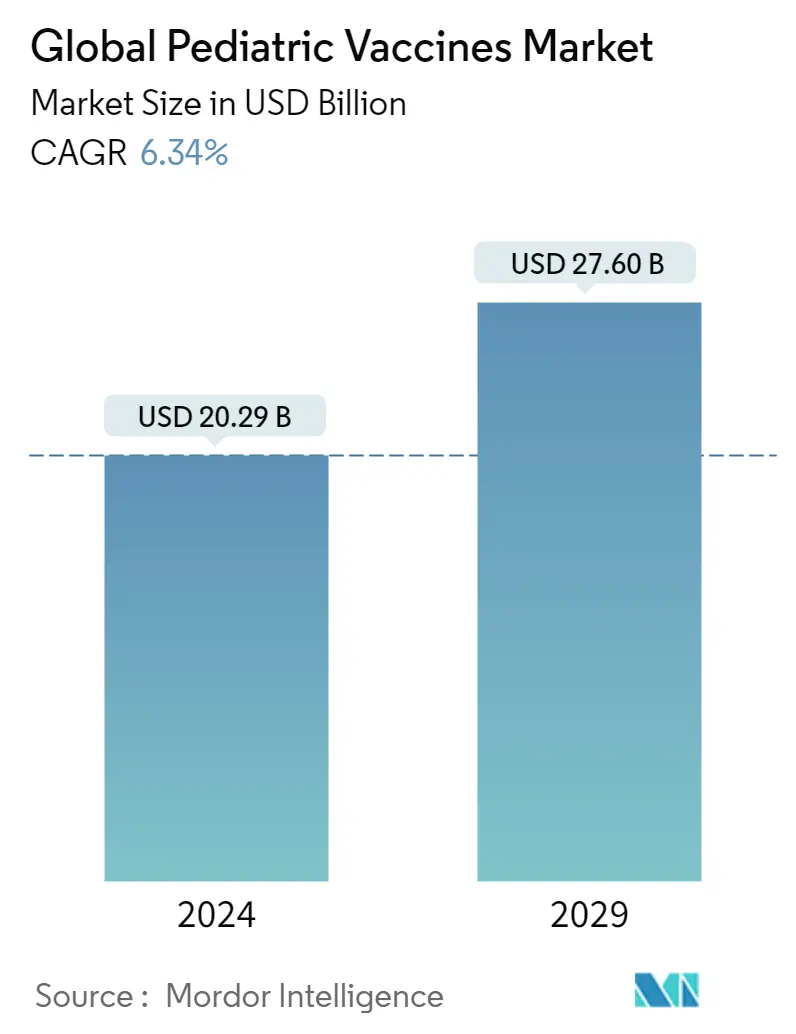
| Study Period | 2019 - 2029 |
| Market Size (2024) | USD 20.29 Billion |
| Market Size (2029) | USD 27.60 Billion |
| CAGR (2024 - 2029) | 6.34 % |
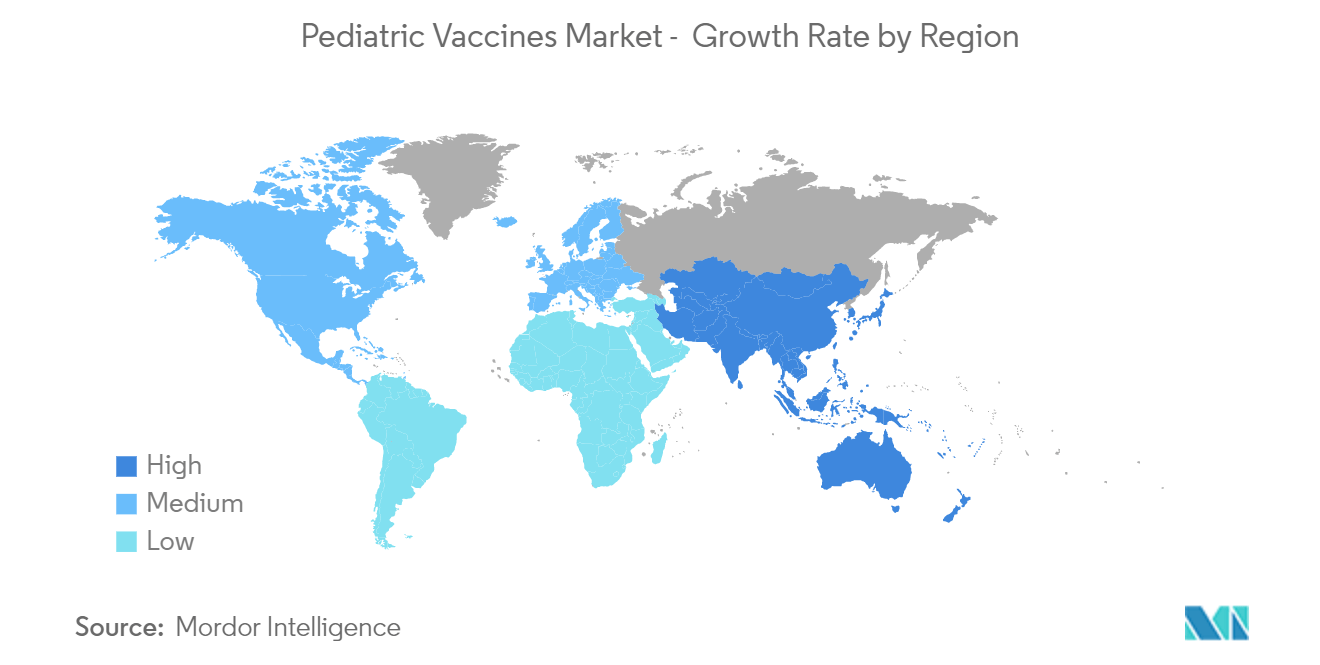
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Pediatric Vaccines Market Analysis
The Global Pediatric Vaccines Market size is estimated at USD 20.29 billion in 2024, and is expected to reach USD 27.60 billion by 2029, growing at a CAGR of 6.34% during the forecast period (2024-2029).
The pediatric vaccine market experienced a severe setback in terms of growth as a result of the abrupt outbreak of COVID-19. As the number of COVID-19 cases surged, healthcare systems redirected their focus to managing the disease, delaying the prevention, diagnosis, and treatment of other chronic illnesses in the pediatric population. According to World Health Organization(WHO) data in 2021, the number of entirely unvaccinated children rose by 3.4 million in 2020. In 2020, only 19 vaccines were introduced, which is less than half of any year in the previous two decades. Moreover, in 2020, 1.6 million female children were not fully immunized against the human papillomavirus (HPV) as the year before. Thus, the impact of the pandemic on the market in the initial phase was adverse. However, due to the relaxation of restrictions and the resumption of R&D activities and clinical trials for pediatric vaccinations, the market is likely to gain traction in the coming years. For instance, the United States Food and Drug Administration (FDA) has approved an emergency use authorization for a Pfizer-BioNTech COVID-19 vaccination for children aged 5 to 11. For this vaccine, two doses are given three weeks apart.
Furthermore, the rising burden of chronic diseases, the necessity and awareness of vaccination, developments in the route of vaccine administration, and an increase in government and non-government financing for research and development are some of the significant drivers propelling the market's growth.
For instance, as per the article titled "Clinical Characteristics, Antimicrobial Resistance, and Risk Factors for Mortality in Pediatric Invasive Pneumococcal Disease in Beijing" published in BMC Infectious Diseases in April 2022, researchers looked at clinical features, antibiotic susceptibility, and risk factors for mortality in children with invasive pneumococcal disease (IPD). The research study concluded that meningitis was the most common clinical manifestation of IPD, and it was commonly linked to death. Since the strains in the PCV13 vaccination would cover the majority of cases, PCV13 should be used more widely. Furthermore, according to World Health Organization (WHO) estimates from March 2022, the measles virus is one of the world's most contagious human diseases, affecting over 100,000 children each year, and rubella is a leading vaccine-preventable cause of birth abnormalities. Only two doses of a safe and efficient vaccine can prevent measles and rubella. Additionally, as per an article titled "Measles" published in January 2022, young children and pregnant women who have not been vaccinated are at significant risk of developing measles. Due to growing levels of vaccination coverage and changes in population immunity at various ages, there has been a recent shift to older children and teenagers. Passive antibody transfer protects young infants who have gained immunity from measles, but as these antibodies diminish, they become more susceptible.
As immunization plays a significant role in disease prevention, there are several international programs for children, which may vary based on the disease. According to World Health Organization (WHO) data published in March 2022, India initiated the Intensified Mission Indradhanush (IMI) 4.0 in phases to meet its objective of providing 90 percent vaccination coverage and beyond to expand the scope of the universal immunization program, which vaccinates nearly 3 crore pregnant women and 2.6 crore children each year. The National Strategic Plan for Achieving and Sustaining Measles and Rubella Elimination in India was adopted , and the MR vaccine campaign immunized nearly 324 million children till 2020. For the first time, India met the global guideline of 2 per 100,000 population for non-measles, non-rubella discard rates in the last six months of 2021. Thus, the aforementioned factors are anticipated to drive the market's growth over the forecast period.
The high cost of immunization and limited access to healthcare in low- and middle-income countries, on the other hand, are expected to hamper the market's growth over the forecast period.
Pediatric Vaccines Market Trends
This section covers the major market trends shaping the Pediatric Vaccines Market according to our research experts:
The Conjugate Vaccine Segment is Expected to Grow at a Significant Rate
Conjugate vaccines consist of antigens (poly or oligosaccharides) that are chemically attached to the carrier protein. Attaching the saccharides to protein converts polysaccharides into T-dependent antigens, which elicit robust immune responses in infants and adults. These "glycoconjugate" vaccines elicit T-cell help for B-cells that produce IgG antibodies to the conjugated polysaccharide. The CDC recommends the routine administration of the pneumococcal conjugate vaccine (PCV13 or Prevnar13) for all children under two years of age.
Furthermore, the rising focus on research, government initiatives on vaccination, and growing awareness, coupled with the companies' efforts in advancing the vaccines, are expected to have a direct correlation with the segment's growth. For instance, in May 2021, Merck's investigational 15-valent pneumococcal conjugate vaccine, V114, showed promising top-line results from two Phase III pediatric trials. Likewise, in July 2021, Tamil Nadu launched the pneumococcal conjugate vaccination as part of the Universal Immunization Program. Every year, 9,23,000 children in the state will benefit from this.
Moreover, in December 2021, India expanded its Pneumococcal Conjugate vaccine coverage to include the PCV13 vaccine in its national childhood immunization program to reduce childhood mortality. In April 2022, the Family Welfare Division of Nepal added the typhoid conjugate vaccine (TCV) to its Expanded Program for Immunization to protect children from typhoid.
Thus, the aforementioned factors together contribute toward segment growth over the forecast period.
The North American Region Holds the Largest Market Share and is Projected to Follow the Same Trend over the Forecast Period
The North American pediatric vaccine market is expected to garner a significant market share. This is primarily due to the well-established healthcare infrastructure, especially in the United States, and a surge in immunization programs. The scheduled child immunization programs under the Centers for Disease Control and Prevention (CDC), high funding, wide commercial availability of vaccines, and society's awareness regarding vaccines contribute to the sustained market growth in the United States.
Considering the factors responsible for the large size of the United States pediatric vaccine market, the country witnesses the highest percentage of children getting vaccinations. This is well described by the data provided by the Kaiser Family Foundation in January 2022, which states that nationally, more than a quarter (28.1%) of 5-to-11-year-olds have received at least one COVID-19 vaccine dose as of January 18, 2022. Moreover, the CDC publishes a recommended child and adolescent immunization schedule along with doses every year for children in the United States. This greatly boosts the adoption of vaccines among the pediatric population, thereby contributing to market growth.
Thus, the abovementioned factors greatly contribute to the market growth in the North American region.
Pediatric Vaccines Industry Overview
The pediatric vaccines market consists of several major players, including Merck & Co. Inc., Sanofi SA, Pfizer Inc., Sinova, and Pfizer, among others. The companies are implementing certain strategic initiatives, such as mergers, new product launches, acquisitions, and partnerships, that help them strengthen their market position.
Pediatric Vaccines Market Leaders
-
Sanofi SA
-
Merck & Co., Inc.
-
Pfizer Inc.
-
Sinovac Biotech Ltd.
-
AstraZeneca plc
*Disclaimer: Major Players sorted in no particular order
Pediatric Vaccines Market News
- In June 2022 Pfizer and BioNTech received the United States Food and Drug Administration received emergency use authorization of the Pfizer-BioNTech COVID-19 vaccine as a three-dose series for children 6 months through 4 years of age.
- In August 2021, Merck published top-line findings from the pivotal PNEU-PED (V114-029) study evaluating the immunogenicity, safety, and tolerability of VAXNEUVANCE (Pneumococcal 15-valent Conjugate Vaccine) in healthy infants enrolled between 42-90 days of age. In the trial, infants were given a 4-dose regimen of either VAXNEUVANCE or the licensed 13-valent pneumococcal conjugate vaccine (PCV13) at 2, 4, 6, and 12-15 months of age.
Pediatric Vaccines Market Report - Table of Contents
1. INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
- 4.1 Market Overview
-
4.2 Market Drivers
- 4.2.1 Rising Burden of Chronic Diseases with the Importance and Awareness of Immunization
- 4.2.2 Increase in the Government and Non-government Funding in R&D
-
4.3 Market Restraints
- 4.3.1 Cost of Immunization
- 4.3.2 Lesser Medical Coverage and Healthcare Services in Low- and Middle-income Countries
-
4.4 Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
-
5.1 By Vaccine Type
- 5.1.1 Monovalent
- 5.1.2 Multivalent
-
5.2 By Technology
- 5.2.1 Live Attenuated
- 5.2.2 Inactivated
- 5.2.3 Toxoid
- 5.2.4 Conjugate
- 5.2.5 Other Technologies
-
5.3 By Application
- 5.3.1 Pneumococcal Disease
- 5.3.2 Influenza
- 5.3.3 Measles, Mumps, and Rubella (MMR)
- 5.3.4 Other Applications
-
5.4 Geography
- 5.4.1 North America
- 5.4.1.1 United States
- 5.4.1.2 Canada
- 5.4.1.3 Mexico
- 5.4.2 Europe
- 5.4.2.1 Germany
- 5.4.2.2 United Kingdom
- 5.4.2.3 France
- 5.4.2.4 Italy
- 5.4.2.5 Spain
- 5.4.2.6 Rest of Europe
- 5.4.3 Asia-Pacific
- 5.4.3.1 China
- 5.4.3.2 Japan
- 5.4.3.3 India
- 5.4.3.4 Australia
- 5.4.3.5 South Korea
- 5.4.3.6 Rest of Asia-Pacific
- 5.4.4 Middle-East and Africa
- 5.4.4.1 GCC
- 5.4.4.2 South Africa
- 5.4.4.3 Rest of Middle-East and Africa
- 5.4.5 South America
- 5.4.5.1 Brazil
- 5.4.5.2 Argentina
- 5.4.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
-
6.1 Company Profiles
- 6.1.1 GlaxoSmithKline PLC
- 6.1.2 Indian Immunologicals Limited
- 6.1.3 AstraZeneca plc
- 6.1.4 Merck & Co. Inc.
- 6.1.5 Mitsubishi Tanabe Pharma Corporation
- 6.1.6 Pfizer Inc.
- 6.1.7 Sanofi SA
- 6.1.8 Seqirus (CSL Limited)
- 6.1.9 Sinovac Biotech Ltd
- 6.1.10 Novavax, Inc.
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
** Subject To AvailablityPediatric Vaccines Industry Segmentation
As per the scope of the report, pediatric vaccines are vaccines that stimulate the body's immune response, in order to prevent a specific disease in children. The Pediatric Vaccines Market is segmented by Vaccine Type (Monovalent and Multivalent), Technology (Live Attenuated, Inactivated, Toxoid, Conjugate, and Other Technologies), Application (Pneumococcal Disease, Influenza, Measles, Mumps, and Rubella (MMR), and Other Applications), and Geography (North America, Europe, Asia-Pacific, Middle-East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions, globally. The report offers the value (USD million) for all the above segments.
| By Vaccine Type | Monovalent | |
| Multivalent | ||
| By Technology | Live Attenuated | |
| Inactivated | ||
| Toxoid | ||
| Conjugate | ||
| Other Technologies | ||
| By Application | Pneumococcal Disease | |
| Influenza | ||
| Measles, Mumps, and Rubella (MMR) | ||
| Other Applications | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Geography | Europe | Germany |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Geography | Asia-Pacific | China |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Geography | Middle-East and Africa | GCC |
| South Africa | ||
| Rest of Middle-East and Africa | ||
| Geography | South America | Brazil |
| Argentina | ||
| Rest of South America |
Pediatric Vaccines Market Research FAQs
How big is the Global Pediatric Vaccines Market?
The Global Pediatric Vaccines Market size is expected to reach USD 20.29 billion in 2024 and grow at a CAGR of 6.34% to reach USD 27.60 billion by 2029.
What is the current Global Pediatric Vaccines Market size?
In 2024, the Global Pediatric Vaccines Market size is expected to reach USD 20.29 billion.
Who are the key players in Global Pediatric Vaccines Market?
Sanofi SA, Merck & Co., Inc. , Pfizer Inc., Sinovac Biotech Ltd. and AstraZeneca plc are the major companies operating in the Global Pediatric Vaccines Market.
Which is the fastest growing region in Global Pediatric Vaccines Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Global Pediatric Vaccines Market?
In 2024, the North America accounts for the largest market share in Global Pediatric Vaccines Market.
What years does this Global Pediatric Vaccines Market cover, and what was the market size in 2023?
In 2023, the Global Pediatric Vaccines Market size was estimated at USD 19.08 billion. The report covers the Global Pediatric Vaccines Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Global Pediatric Vaccines Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Pediatric Vaccines Industry Report
Statistics for the 2023 Pediatric Vaccines market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Pediatric Vaccines analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.



