Market Trends of Pharmacovigilance and Drug Safety Software Industry
This section covers the major market trends shaping the Pharmacovigilance & Drug Safety Software Market according to our research experts:
Fully Integrated Software Expected to Witness Significant Growth Over the Forecast Period
Monitoring and tracking an organization's drug safety reporting process can help ensure that issues are reported and addressed as soon as they happen, preventing delays and/or false reporting, which can lead to regulatory non-compliance, monetary fines, and unwanted attention.
The issue-tracking software allows for electronic reporting of suspected adverse drug reactions and effective data analysis. This allows for the early discovery of safety issues. Vertical growth is projected to be aided by a strong foothold of key market players operating in the field of issue-tracking software. For instance, in July 2021, Chemotargets launched CLARITY PV, a new web-based platform for translational safety and pharmacovigilance purposes. CLARITY PV platform contains highly-curated structured and integrated data covering the entire safety lifetime of a drug, from safety pharmacology, through preclinical toxicology and clinical safety stages, up to post-marketing reports, which allows a seamless connection and traceability of safety signals across all drug discovery phases.
Moreover, with the COVID-19 pandemic, many companies launched new issue-tracking software to better serve patients. For instance, in May 2021, Dialog Solutions introduced the next generation of pharmacovigilance literature monitoring: Drug Safety Triager. Thus, the launch of such a product may work in the favor of segment growth.
The increasing incidence of adverse drug reactions (ADR) is expected to propel the demand for AER software in the coming years.
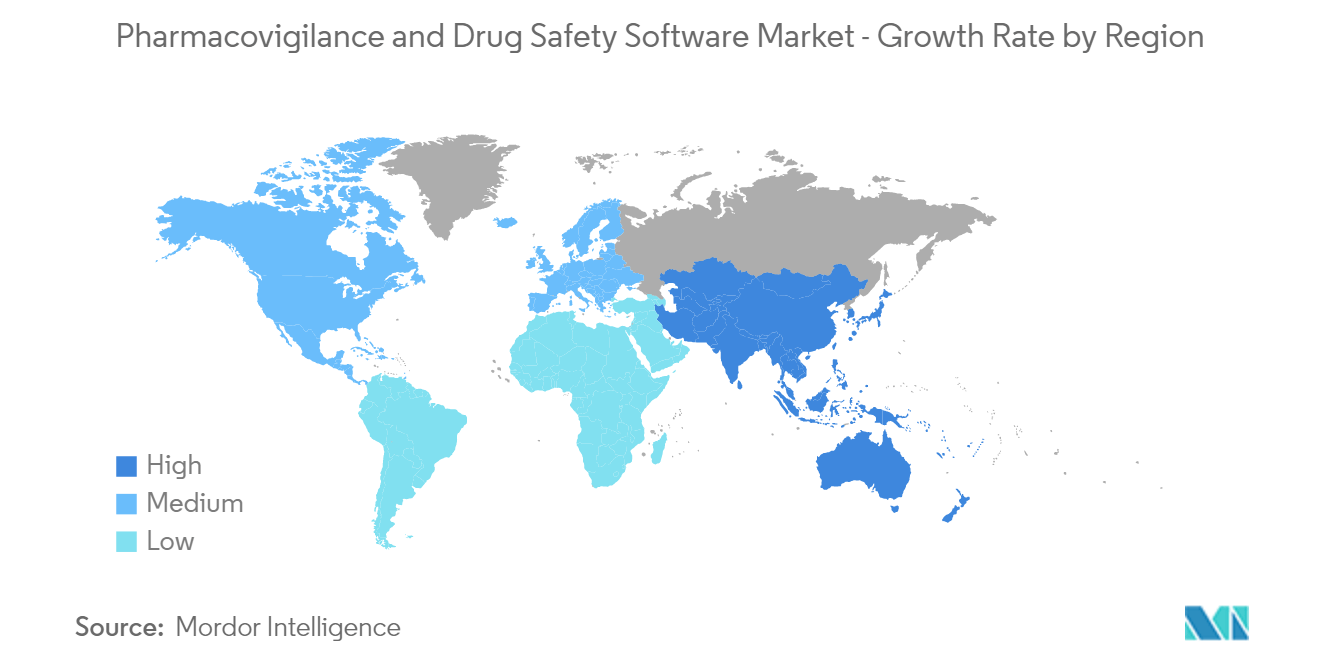
North America is Expected to Witness A healthy Growth Over the Forecast Period
North America is expected to experience healthy growth in the pharmacovigilance and drug safety software market due to the increasing research expenditure and government initiatives. Adverse drug reactions are one of the major causes of hospitalizations and deaths in the United States, thus fueling the pharmacovigilance and drug safety software market. For instance, in 2021, monitoring by the Vaccine Adverse Event Reporting System detected 10 cases of anaphylaxis after administration of a reported 4,041,396 first doses of Moderna COVID-19 vaccine (2.5 cases per million doses). Thus, the regulatory authorities' growing need for medical information is also anticipated to fuel this segment's growth.
Initiatives undertaken by the governments are also propelling the market's growth in North America. For instance, the Open FDA initiative undertaken by the US government provides access to its database by open search-based programs for application developers and scientists. The United States initiated another project called Mini-Sentinel to promote an active surveillance system by providing relevant statistical data in less time.
Moreover, in October 2021, in response to the pandemic, the Food and Drug Administration of the United States introduced FDA Adverse Event Reporting System (FAERS), a dashboard for COVID-19 emergency use authorization (EUA) products. The tool provided updates of adverse event reports submitted for drugs under emergency use authorization for COVID-19.
Therefore, owing to the aforesaid factors, the growth of the studied market was anticipated in the North American region