Market Trends of Preclinical CRO Industry
The Toxicology Testing Segment is Predicted to Witness Significant Growth Between 2024 and 2029
Toxicology testing involves the evaluation of the potential adverse effects of chemical substances or pharmaceutical compounds on living organisms. It encompasses a range of methodologies to assess these substances' safety profile, including their potential to cause harm or toxicity. In the dynamic landscape of pharmaceutical research and development, the demand for preclinical toxicology testing services offered by contract research organizations (CROs) is witnessing a significant upsurge. This surge can be attributed to various factors, including the increasing collaboration between pharmaceutical companies and CROs and technological advancements enhancing the efficiency and accuracy of toxicology testing processes.
The expanding partnership between pharmaceutical companies and CROs drives the demand for toxicology testing services. As pharmaceutical companies intensify their drug development efforts, they seek CROs' expertise and infrastructure to conduct essential preclinical toxicology studies. For instance, in May 2023, Immuter partnered with Charles River Laboratories to conduct a good laboratory practice (GLP) toxicology study for its proprietary candidate IMP761 to address autoimmune diseases. Similarly, in March 2024, Badvus Research forged a strategic alliance with Southern Research, focusing on preclinical toxicology testing and regulatory approval processes. These collaborations underscore the pivotal role of CROs in supporting drug development initiatives and ensuring regulatory compliance through comprehensive toxicology assessments.
Toxicology testing in preclinical CROs serves as a vital component of the drug development process, enabling pharmaceutical companies to evaluate the safety profiles of their candidates and make informed decisions regarding their progression in the development pipeline. Hence, increasing the vertical collaboration between pharma companies and CROs is likely to enhance the segmental growth of the market.
North America is Expected to Hold a Significant Share of the Market Between 2024 and 2029
The North American preclinical CRO market is expected to benefit from higher in-house drug development and discovery costs, rising prevalence of chronic diseases, increasing complexity in clinical trials, and a high number of investigational candidates in the development pipeline.
The higher prevalence of chronic diseases like cardiovascular diseases, diabetes, cancer, and neurological disorders necessitates the development of new therapies and treatments. Such a high and evolving burden of chronic diseases forces pharma companies to outsource preclinical trials to investigate promising candidates against those diseases, which is projected to foster the country’s market growth over the study period. For instance, according to the American Cancer Society, the prevalence of lung cancer is increasing in the country, and the country reported 238,340 lung cancer cases in 2023 compared to 236,740 in 2022. This data shows a rapid increase in the incidence of cancer cases in the country, and between 2024 and 2029, the incidence of cancer will further increase; thus, the escalating burden of cancer cases in the country is projected to spur the demand for advanced therapeutics and which is in turn projected to foster preclinical trial activities and is anticipated to support country’s market growth over the study period.
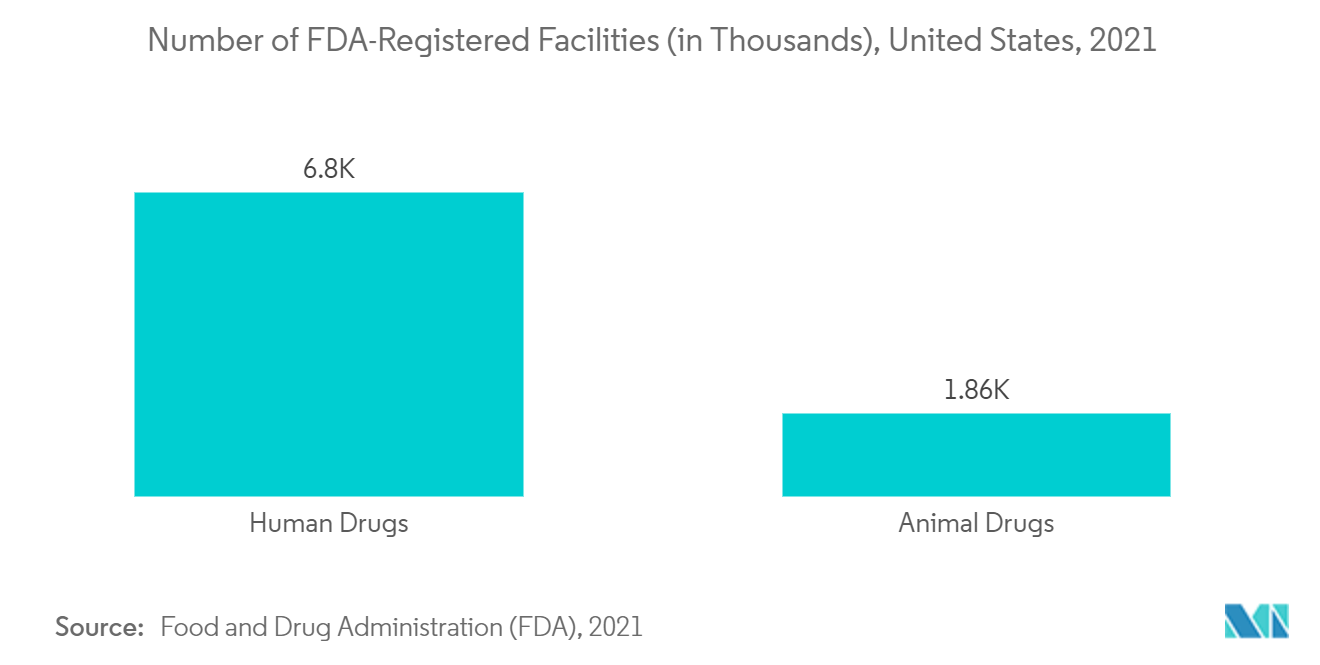
In addition, the higher in-house drug discovery and development costs facilitate pharmaceutical companies to outsource preclinical trial activities to reduce the cost burden and increased complexity associated with clinical trials. Thus, drug discovery and development costs are anticipated to foster demand for preclinical contract research organizations (CROs) to conduct clinical trials as they have all the required infrastructures. For instance, according to a study conducted by the World Health Organization (WHO) in 2022, the average cost to develop a new drug ranges from USD 43.4 million to USD 4.2 million. Thus, the significant cost associated with new drug development is expected to facilitate the demand for preclinical CROs to perform clinical trials and other research activities on behalf of pharmaceutical companies, as CROs have a better experience and infrastructure to conduct research services. Thus, the high demand for preclinical CROs to conduct research activities is projected to support the country’s market growth over the study period.
Hence, the high prevalence of chronic diseases and the higher in-house drug discovery and development costs are expected to drive the studied market in North America between 2024 and 2029.