Small Molecules Innovator Contract Development And Manufacturing Organization Market Size
| Study Period | 2021 - 2029 |
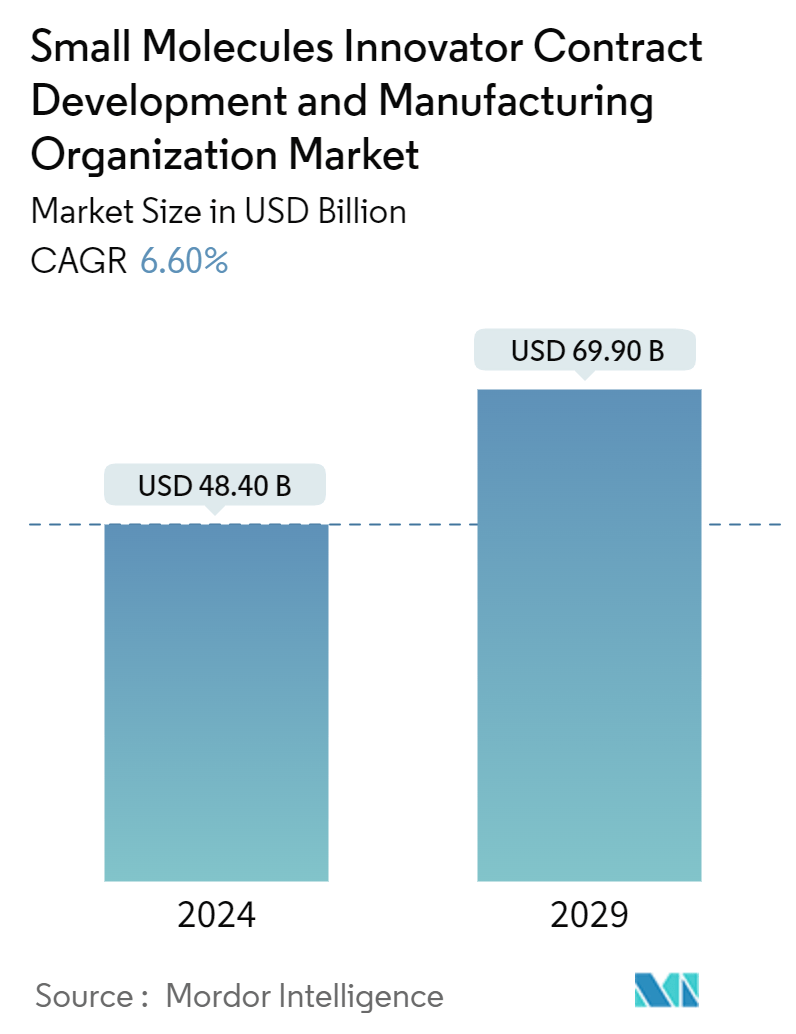
| Market Size (2024) | USD 48.40 Billion |
| Market Size (2029) | USD 69.90 Billion |
| CAGR (2024 - 2029) | 6.60 % |
| Fastest Growing Market | North America |
| Largest Market | Asia Pacific |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Small Molecules Innovator Contract Development And Manufacturing Organization Market Analysis
The Small Molecules Innovator Contract Development And Manufacturing Organization Market size is estimated at USD 48.40 billion in 2024, and is expected to reach USD 69.90 billion by 2029, growing at a CAGR of 6.60% during the forecast period (2024-2029).
The market is driven by increasing demand for small molecule drugs, the growing pipeline of small molecule drugs, the increasing burden of chronic diseases, and rising pharmaceutical R&D investments. A robust pipeline of small molecule drugs means that pharmaceutical and biotech companies have more compounds in various stages of development. This creates a greater need for specialized services CDMOs provide, including formulation development, process optimization, and manufacturing. For instance, according to the Bio Industry Analysis Report published in February 2023, 47 small molecule new chemical entities (NCEs) were in the clinical pipeline as of 2022. Traditional systemic small-molecule antibiotics account for 97%, and topical small molecules account for 3% of the small-molecule pipeline NCEs.
Similarly, according to Clinicaltrials.gov, as of March 2024, there were 64 small molecules of drugs in phase II trials and 3 in the phase III trials. Hence, the growing pipeline often spans various therapeutic areas, addressing multiple medical conditions. This diversification in drug development increases the demand for CDMOs with expertise in different fields, further stimulating the market.
The substantial investment by pharmaceutical companies in research and development (R&D) of small molecule drugs has led to an increased demand for the services provided by contract development and manufacturing organizations (CDMOs). These organizations require specialized expertise and infrastructure to bring their drug candidates from the discovery phase through clinical trials. For instance, in September 2022, Cambrex completed the initial stage of its USD 30 million investment in its High Point, North Carolina facility for manufacturing small molecule active pharmaceutical ingredients (APIs). Furthermore, strategic collaboration for small molecule drug development is expected to drive market growth over the projected period. For instance, in January 2022, Sanofi and Exscientia entered a strategic research alliance and licensing pact for the purpose of developing up to 15 novel small molecule candidates across oncology and immunology, leveraging Exscientia’s AI-driven platform utilizing actual patient samples.
Hence, the strong pipeline for small molecule drugs, increasing investment in developing small molecules, and key adoption of key strategies by market participants are expected to drive segment growth. However, stringent government regulations and compliance issues with outsourcing are anticipated to restrain the market growth over the projected period.
Small Molecules Innovator Contract Development And Manufacturing Organization Market Trends
The Neurology Segment is Expected to Hold Significant Share Between 2024 and 2029
The neurology segment in the small molecule innovators contract development and manufacturing organization (CDMO) market has been experiencing notable growth owing to growing awareness and understanding of neurological disorders, leading to an increased emphasis on the development of drugs targeting conditions like Alzheimer's, Parkinson's, epilepsy, and various other neurodegenerative diseases.
For instance, in July 2023, Biogen announced its small molecules pipeline drug Zuranolone (GABAA PAM) – Major depressive disorder (MDD) in phase III trials and BIIB131 (plasminogen activator) – Acute ischemic stroke in Phase II Trials. Similarly, UCB SA also reported that it has 4 small molecules of innovative drugs in the pipeline for neurology diseases, which include fenfluramine (5-HT agonist), doxecitine and doxribtimine (MT1621, nucleoside therapy), minzasolmin (α-syn-misfolding inhibitor), and STACCATO alprazolam (benzodiazepine) in various phase of development. Such a huge pipeline for small molecule innovator drugs is expected to create a huge demand for CDMO for process development, analytical testing, and manufacturing, which is expected to contribute to segment growth between 2024 and 2029.
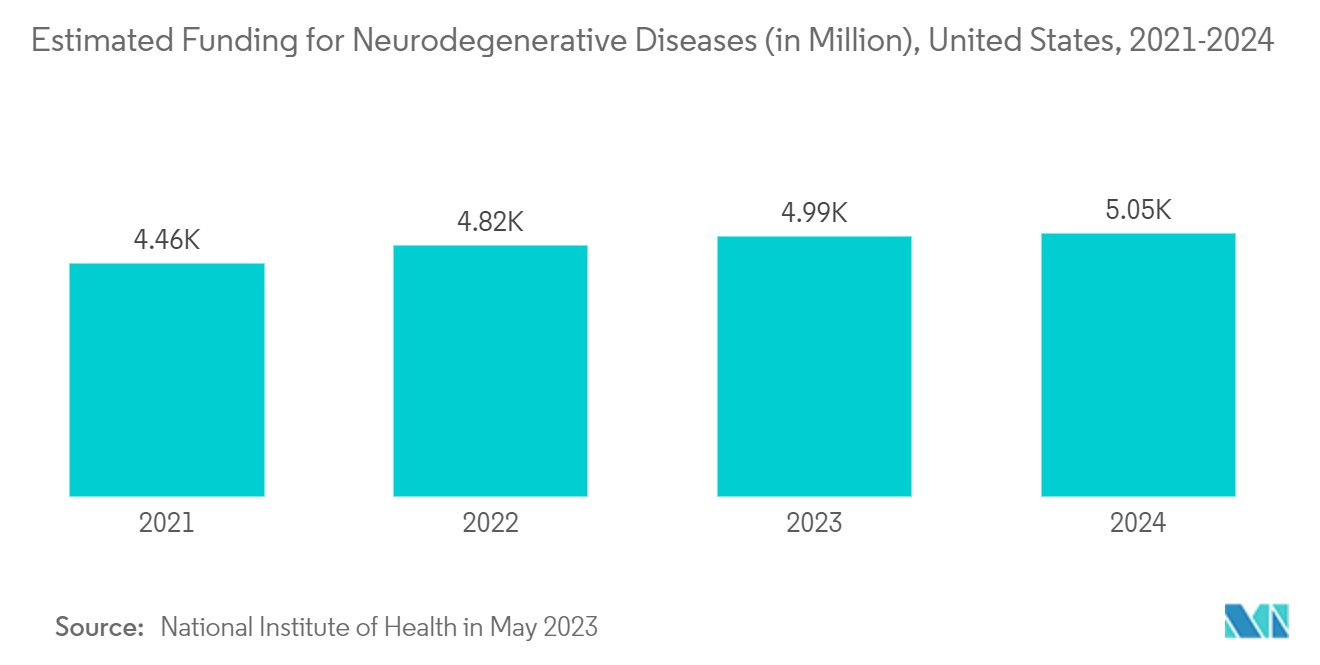
An increase in CNS diseases, like Parkinson's disease, amyotrophic lateral sclerosis (ALS), Huntington chorea, and Alzheimer's disease, facilitates the demand for CRO services since the drug development of these diseases requires various clinical trial support, regulatory recommendations, etc. For instance, according to data published by Alzheimer’s Disease Facts and Figures Annual Report 2023, over 6 million Americans suffered from Alzheimer's disease, a figure predicted to climb to almost 13 million within three decades.
Additionally, as per the above source, in 2023, the United States spent USD 345 billion in treating and managing Alzheimer’s and other dementias. By 2050, these costs could rise to nearly USD 1 trillion. Thus, the high burden of the disease is creating demand for innovative and effective therapies, driving the growth of the studied market.
Furthermore, the data updated by the World Health Organization in March 2023 showed that globally, about 55 million people suffer from dementia; every year, there are nearly 10 million new cases of dementia filed. Additionally, Atlas of Multiple Sclerosis data updated in September 2023 showed that 2.9 million people live with multiple sclerosis in 2023 worldwide and 10 million with Parkinson's disease, as per the data published by the Parkinson's Foundation. These increasing burdens of CNS disorders are anticipated to drive the demand for central nervous system therapeutics and significant research activities between 2024 and 2029.
Pharmaceutical and biotechnology corporations are augmenting their focus on service expansion. For instance, in September 2023, Cellectricon expanded its neuroscience contract research portfolio with a new Neuroplasticity Services launched module set to accelerate drug discovery in therapeutic areas such as neuropsychiatry, epilepsy, and neurodegeneration.
Hence, increasing research and development activities, the presence of a strong pipeline, and strategic activities by the market players are expected to contribute to market growth between 2024 and 2029.
North America is Expected to Hold a Significant Market Share Between 2024 and 2029
North America, particularly the United States, is home to a well-established and robust pharmaceutical industry. This region has a high concentration of innovative pharmaceutical and biotech companies that are actively engaged in drug discovery and development. The high investment in R&D, the strong foothold of key market players, and rising grants from the National Institute of Health for developing novel therapeutics in the country also contribute to market growth.
The increasing investment by the pharmaceutical and biotechnology industry in the development of small-molecule drugs is expected to contribute to market growth. For instance, in May 2023, PharmEnable announced it had closed a Pre-Series A investment round of USD 7.5 million to develop the next generation of small molecule drugs against disease areas of high clinical need. Further, market players are engaging in service expansion, and collaboration is likely to propel the growth of the market. For instance, in April 2023, Phlow, a CDMO, announced that it had sealed USD 36 million through a Series B capital raise. Phlow stated that it would use the capital to expand its commercial offerings, which includes growing its CDMO program, called 'cdmoX. Additionally, in July 2022, Alliance Pharma, a company providing small and large-molecule bioanalytical services in the pharmaceutical and biopharmaceutical industry, closed the purchase of Drug Development Solutions (DDS) from LGC. Ampersand Capital Partners and KKR & Co. Inc.
Hence, increasing investment and strategic activities by the market players are expected to boost the market in the region between 2024 and 2029.
Small Molecules Innovator Contract Development And Manufacturing Organization Industry Overview
The small molecules innovator contract development and manufacturing organization market is fragmented in nature. In order to strengthen their position in the market, companies are implementing noteworthy strategies and measures to expand their industry presence. Some of the key market players are Eurofins Scientific, Cambrex Corporation, Catalent, Thermo Fisher Scientific Inc., and Jubilant Pharmova Limited.
Small Molecules Innovator Contract Development And Manufacturing Organization Market Leaders
-
Eurofins Scientific
-
Cambrex Corporation
-
Catalent
-
Thermo Fisher Scientific Inc.
-
Jubilant Pharmova Limited
*Disclaimer: Major Players sorted in no particular order
Small Molecules Innovator Contract Development And Manufacturing Organization Market News
- June 2023: Porton Pharma Solutions established a state-of-the-art Small Molecule Platform in Shanghai to revolutionize CDMO services. Porton Pharma Solutions Ltd now has seven R&D and production facilities in Shanghai that are equipped to provide comprehensive solutions for drug substances, drug preparation, synthetic macromolecules, and biological macromolecules.
- May 2023: GeneOne Life Science Inc., a CDMO, and the Wistar Institute announced a collaboration to identify novel small molecules capable of inhibiting Nipah virus entry into cells and to develop these into preclinical leads for future advancement into global Phase I clinical trials for the treatment and/or post-exposure prophylaxis of Nipah virus infection.
Small Molecules Innovator Contract Development And Manufacturing Organization Market Report - Table of Contents
1. INTRODUCTION
- 1.1 Study Assumptions and Market Definitions
- 1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
- 4.1 Market Overview
-
4.2 Market Drivers
- 4.2.1 Increasing Demand for Small Molecule Drugs and Growing Pipeline of Small Molecule Drugs
- 4.2.2 Growing Burden of Chronic Diseases
- 4.2.3 Increasing Pharmaceutical R&D Investments
-
4.3 Market Restraints
- 4.3.1 Stringent Government Regulations
- 4.3.2 Compliance Issues with Outsourcing
-
4.4 Porter's Five Forces Analysis
- 4.4.1 Bargaining Power of Suppliers
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Threat of New Entrants
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value – USD)
-
5.1 By Product
- 5.1.1 Small Molecule API
- 5.1.2 Small Molecule Drug Product
- 5.1.2.1 Oral solid dose
- 5.1.2.2 Semi-Solid Dose
- 5.1.2.3 Liquid Dose
- 5.1.2.4 Others
-
5.2 By Stage
- 5.2.1 Preclinical
- 5.2.2 Clinical
- 5.2.2.1 Phase I
- 5.2.2.2 Phase II
- 5.2.2.3 Phase III
- 5.2.2.4 Phase IV
- 5.2.3 Commercial
-
5.3 By End User
- 5.3.1 Pharmaceutical and Biotechnology
- 5.3.2 Contract Research Organization
-
5.4 By Therapeutic Area
- 5.4.1 Cardiovascular disease
- 5.4.2 Oncology
- 5.4.3 Respiratory disorders
- 5.4.4 Neurology
- 5.4.5 Metabolic disorders
- 5.4.6 Infectious disease
- 5.4.7 Others
-
5.5 Geography
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 United Kingdom
- 5.5.2.2 Germany
- 5.5.2.3 France
- 5.5.2.4 Spain
- 5.5.2.5 Italy
- 5.5.2.6 Rest of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 India
- 5.5.3.2 Japan
- 5.5.3.3 China
- 5.5.3.4 Australia
- 5.5.3.5 South Korea
- 5.5.3.6 Rest of Asia-Pacific
- 5.5.4 Middle East and Africa
- 5.5.4.1 GCC
- 5.5.4.2 South Africa
- 5.5.4.3 Rest of the Middle East and Africa
- 5.5.5 South America
- 5.5.5.1 Brazil
- 5.5.5.2 Argentina
- 5.5.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
-
6.1 Company Profiles
- 6.1.1 Eurofins Scientific
- 6.1.2 Cambrex Corporation
- 6.1.3 Catalent
- 6.1.4 Thermo Fisher Scientific Inc.
- 6.1.5 Jubilant Pharmova Limited
- 6.1.6 Lonza Group Ltd
- 6.1.7 Wuxi AppTec
- 6.1.8 Syngene International Limited
- 6.1.9 Almac Group
- 6.1.10 Piramal Pharma Solutions
- 6.1.11 Recipharm AB
- 6.1.12 Labcorp Drug Development
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
** Subject To AvailablitySmall Molecules Innovator Contract Development And Manufacturing Organization Industry Segmentation
As per the scope of the report, the contract development and manufacturing organization (CDMO) market for small molecule innovators refers to companies that provide services to pharmaceutical and biotechnology companies for the development and manufacturing of small molecule drugs. These services may include drug formulation, process development, analytical testing, and manufacturing of clinical trial supplies.
The small molecule contract development and manufacturing organization market is segmented by product, stage type, end user, therapeutic area, and geography. By product, the market is segmented into small molecule API and small molecule drug products. By small molecule drug products, the market is further segmented into oral solid dose, semi-solid dose, liquid dose, and others. By stage type, the market is segmented by preclinical, clinical, and commercial. By end user, the market is segmented by pharmaceutical and biotechnology. By therapeutic area, the market is segmented into cardiovascular disease, oncology, respiratory disorders, neurology, metabolic disorders, infectious disease, and others). By geography, the market is segmented into North America, Europe, Asia-Pacific, Middle East and Africa, and South America. The report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD) for the above segments.
| By Product | Small Molecule API | |
| Small Molecule Drug Product | Oral solid dose | |
| Semi-Solid Dose | ||
| Liquid Dose | ||
| Others | ||
| By Stage | Preclinical | |
| Clinical | Phase I | |
| Phase II | ||
| Phase III | ||
| Phase IV | ||
| Commercial | ||
| By End User | Pharmaceutical and Biotechnology | |
| Contract Research Organization | ||
| By Therapeutic Area | Cardiovascular disease | |
| Oncology | ||
| Respiratory disorders | ||
| Neurology | ||
| Metabolic disorders | ||
| Infectious disease | ||
| Others | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Geography | Europe | United Kingdom |
| Germany | ||
| France | ||
| Spain | ||
| Italy | ||
| Rest of Europe | ||
| Geography | Asia-Pacific | India |
| Japan | ||
| China | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Geography | Middle East and Africa | GCC |
| South Africa | ||
| Rest of the Middle East and Africa | ||
| Geography | South America | Brazil |
| Argentina | ||
| Rest of South America |
Small Molecules Innovator Contract Development And Manufacturing Organization Market Research FAQs
How big is the Small Molecules Innovator Contract Development And Manufacturing Organization Market?
The Small Molecules Innovator Contract Development And Manufacturing Organization Market size is expected to reach USD 48.40 billion in 2024 and grow at a CAGR of 6.60% to reach USD 69.90 billion by 2029.
What is the current Small Molecules Innovator Contract Development And Manufacturing Organization Market size?
In 2024, the Small Molecules Innovator Contract Development And Manufacturing Organization Market size is expected to reach USD 48.40 billion.
Who are the key players in Small Molecules Innovator Contract Development And Manufacturing Organization Market?
Eurofins Scientific, Cambrex Corporation, Catalent, Thermo Fisher Scientific Inc. and Jubilant Pharmova Limited are the major companies operating in the Small Molecules Innovator Contract Development And Manufacturing Organization Market.
Which is the fastest growing region in Small Molecules Innovator Contract Development And Manufacturing Organization Market?
North America is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Small Molecules Innovator Contract Development And Manufacturing Organization Market?
In 2024, the Asia Pacific accounts for the largest market share in Small Molecules Innovator Contract Development And Manufacturing Organization Market.
What years does this Small Molecules Innovator Contract Development And Manufacturing Organization Market cover, and what was the market size in 2023?
In 2023, the Small Molecules Innovator Contract Development And Manufacturing Organization Market size was estimated at USD 45.21 billion. The report covers the Small Molecules Innovator Contract Development And Manufacturing Organization Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the Small Molecules Innovator Contract Development And Manufacturing Organization Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Small Molecules Innovator Contract Development And Manufacturing Organization Industry Report
Statistics for the 2024 Small Molecules Innovator Contract Development And Manufacturing Organization market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Small Molecules Innovator Contract Development And Manufacturing Organization analysis includes a market forecast outlook for 2024 to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
Small Molecules Innovator Contract Development And Manufacturing Organization Market Report Snapshots
- Small Molecules Innovator Contract Development And Manufacturing Organization Market Size
- Small Molecules Innovator Contract Development And Manufacturing Organization Market Share
- Small Molecules Innovator Contract Development And Manufacturing Organization Market Trends
- Small Molecules Innovator Contract Development And Manufacturing Organization Companies



