Sterile Injectable Contract Manufacturing Market Size
| Study Period | 2021 - 2029 |
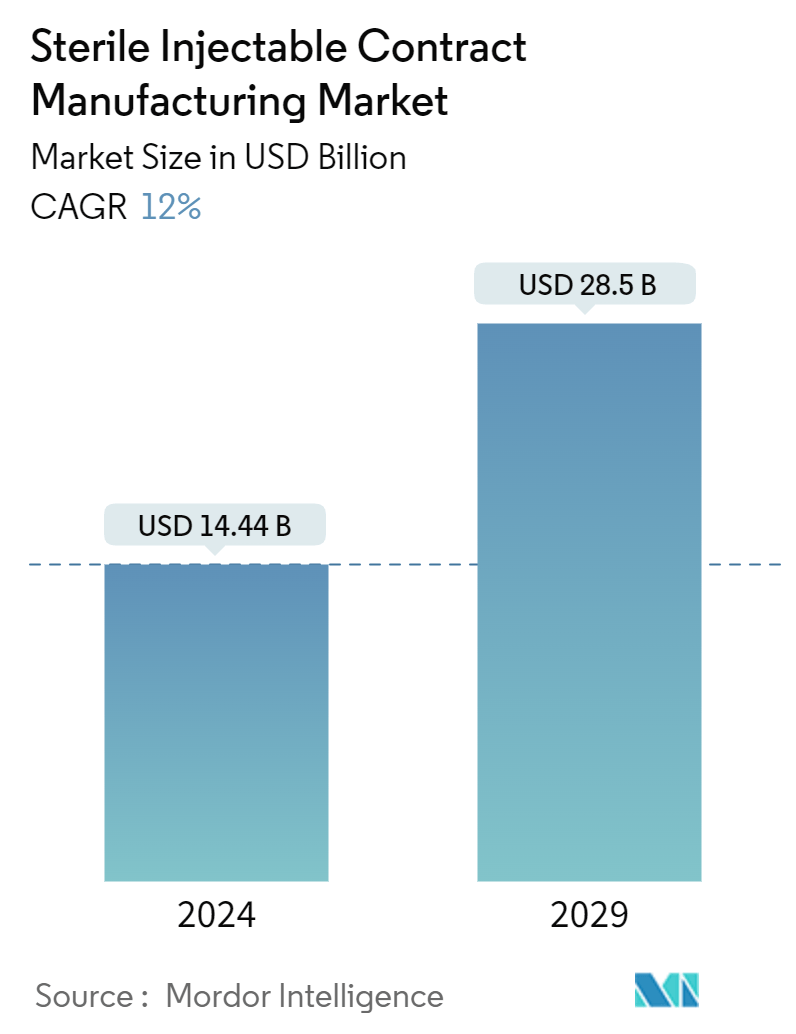
| Market Size (2024) | USD 14.44 Billion |
| Market Size (2029) | USD 28.5 Billion |
| CAGR (2024 - 2029) | 12.00 % |
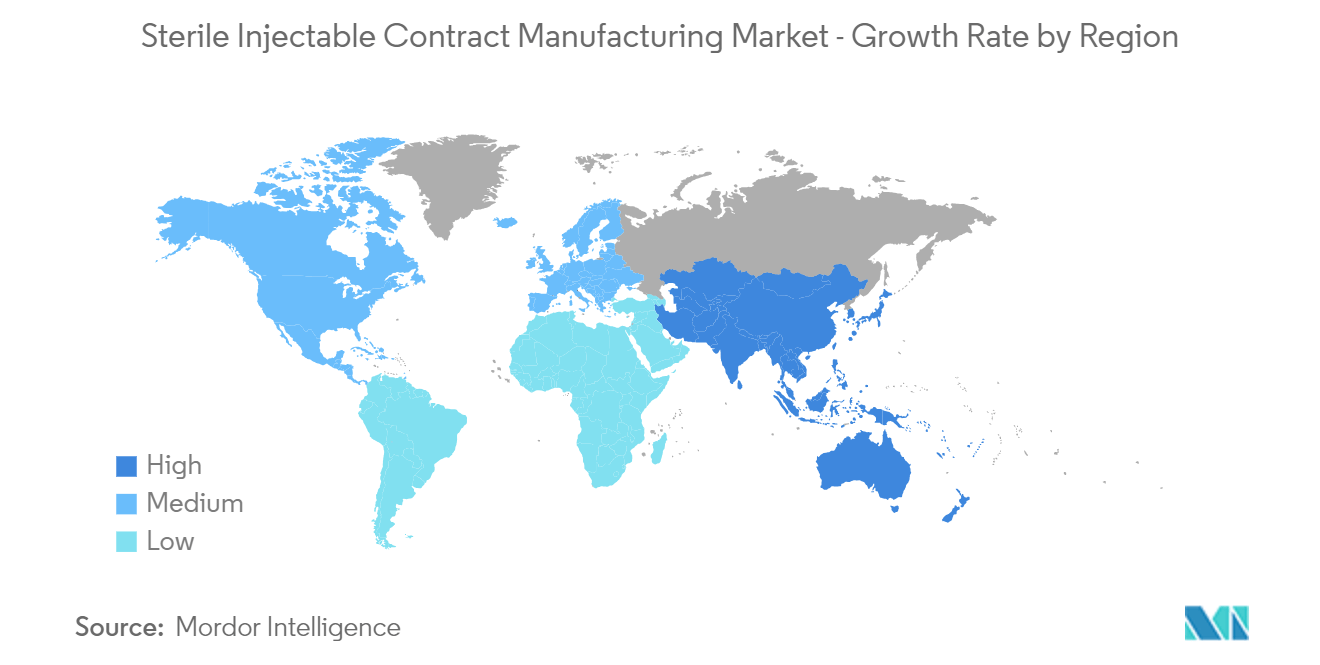
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Sterile Injectable Contract Manufacturing Market Analysis
The Sterile Injectable Contract Manufacturing Market size is estimated at USD 14.44 billion in 2024, and is expected to reach USD 28.5 billion by 2029, growing at a CAGR of 12% during the forecast period (2024-2029).
The increasing pipeline and approvals of injectable drugs are significant drivers of the sterile injectable contract manufacturing market. Pharmaceutical companies are developing injectable drugs due to their advantages, including faster onset of action, precise dosing, and improved patient compliance. For instance, according to the FDA report published in January 2023, CDER approved 37 novel drugs, either as new molecular entities (NMEs) under New Drug Applications (NDAs) or as new therapeutic biological products under Biologics License Applications (BLAs) in 2022. These drugs include injectables, and hence, a strong pipeline is expected to increase the demand for contract manufacturing for commercial and research purposes, thereby contributing to market growth.
The burgeoning demand for cellular and genetic treatments propels the expansion of the market. As the pipeline of these therapies expands, there is a growing need for specialized manufacturing capabilities, which contract manufacturing organizations (CMOs). For instance, according to ClinicalTrials.gov, as of March 2024, more than 581 clinical trials were investigating the potential of cell therapies and related approaches for treating various diseases in North America. Therefore, the high amount of research in clinical development for cell-based therapy is expected to boost contract manufacturing over the forecast period.
Moreover, strategic activities by the market players, such as the expansion of sterile manufacturing facilities, collaboration, and partnership, are expected to contribute to the market growth over the forecast period. For instance, in November 2023, private equity firm NorthEdge invested in a Torbay Pharmaceuticals sterile injectable contract manufacturer and license holder to support its next phase of international growth after acquiring it from Torbay and Devon NHS Foundation Trust. In March 2023, PCI Pharma Services unveiled plans for a USD 50 million expansion at its Rockford, Illinois, sterile injectables site. The project will add a 200,000-square-foot facility to boost the plant’s capacity for injectable drug-device combination products.
Hence, the growing demand for cell and gene therapy in the product pipeline and the expansion of sterile manufacturing facilities are expected to boost market growth over the forecast period. However, challenges related to quality control and high operational costs are anticipated to restrain the market’s growth.
Sterile Injectable Contract Manufacturing Market Trends
The Cancer Segment is Expected to Hold a Significant Share Over the Forecast Period
Cancer therapies necessitate meticulous manufacturing techniques that guarantee drug stability and effectiveness. Sterile injectable contract manufacturers with expertise in handling complex formulations and biologics are well-positioned to meet the unique challenges of oncology drugs. Many cancer treatments involve complex biologics and novel therapeutic approaches. Sterile injectables provide a reliable and effective means of delivering these advanced therapies, contributing to the expansion of the sterile injectable contract manufacturing market.
The Canadian Cancer Society's (CCS) 2023 report highlighted that cancer poses a significant healthcare burden in Canada, with its incidence steadily rising. Thus, the high burden of cancer is creating opportunities for new sterile injectables CMO in the country, driving the segment’s growth. The same source stated that 239,100 cancer cases were reported in Canada in 2023. Thus, the growing burden of cancer is expected to create demand for contract manufacturing services, likely contributing to segmental growth.
The robust growth in outsourcing practices is projected to fuel segment expansion throughout the forecast period. Pharmaceutical companies, including those focused on oncology, are increasingly outsourcing manufacturing activities to streamline operations, reduce costs, and leverage the specialized capabilities of contract manufacturers. This trend has contributed to the growth of the sterile injectable contract manufacturing market within the cancer segment.
Hence, growing cancer prevalence and the increasing trend of outsourcing are expected to boost the segment’s growth over the forecast period.
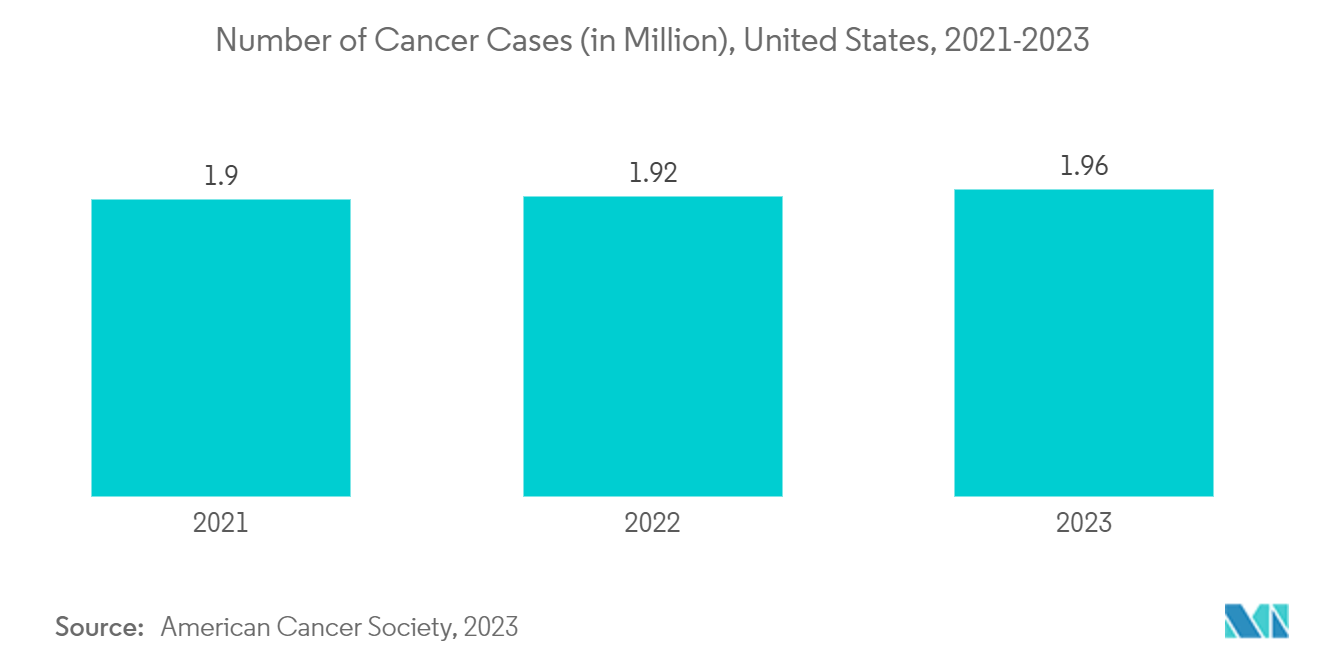
North America is Expected to Hold a Significant Market Share Over the Forecast Period
In North America, the sterile injectable contract manufacturing market is expected to grow due to the establishment of contract manufacturing facilities, the rise in chronic diseases, and the development of advanced biologics. Sterile injectables are crucial for delivering complex therapies, and contract manufacturing provides a scalable solution for meeting this demand.
The increasing emphasis on biologics and biosimilars, which often require sterile injectable formulations, contributes significantly to the growth of the contract manufacturing market. For instance, according to a report by the Food and Drug Administration published in March 2023, 15 biologics were approved in 2022; of this, six were indicated for the treatment of a diversity of cancers. An anticipated surge in biologics drug approvals is projected to boost market growth during the forecast period.
Strategic activities by the market players, such as manufacturing facility expansion, collaboration, partnerships, and acquisitions, are expected to boost the market’s growth over the forecast period. For instance, in October 2023, Sharp acquired Berkshire Sterile Manufacturing (BSM), a contract development and manufacturing organization (CDMO) in Massachusetts that offers clinical and commercial sterile injectable products. Additionally, in March 2022, Sandoz Canada announced the completion of the sale of its Boucherville, Quebec, manufacturing plant to Delpharm, a CMO. This plant is Canada's largest sterile injectable production facility and provides strategic and potentially lifesaving medicines to the Canadian and US healthcare systems, primarily hospitals.
Thus, collaborations with or outsourcing manufacturing to CMOs in the region contribute to the overall growth of the market.
Sterile Injectable Contract Manufacturing Industry Overview
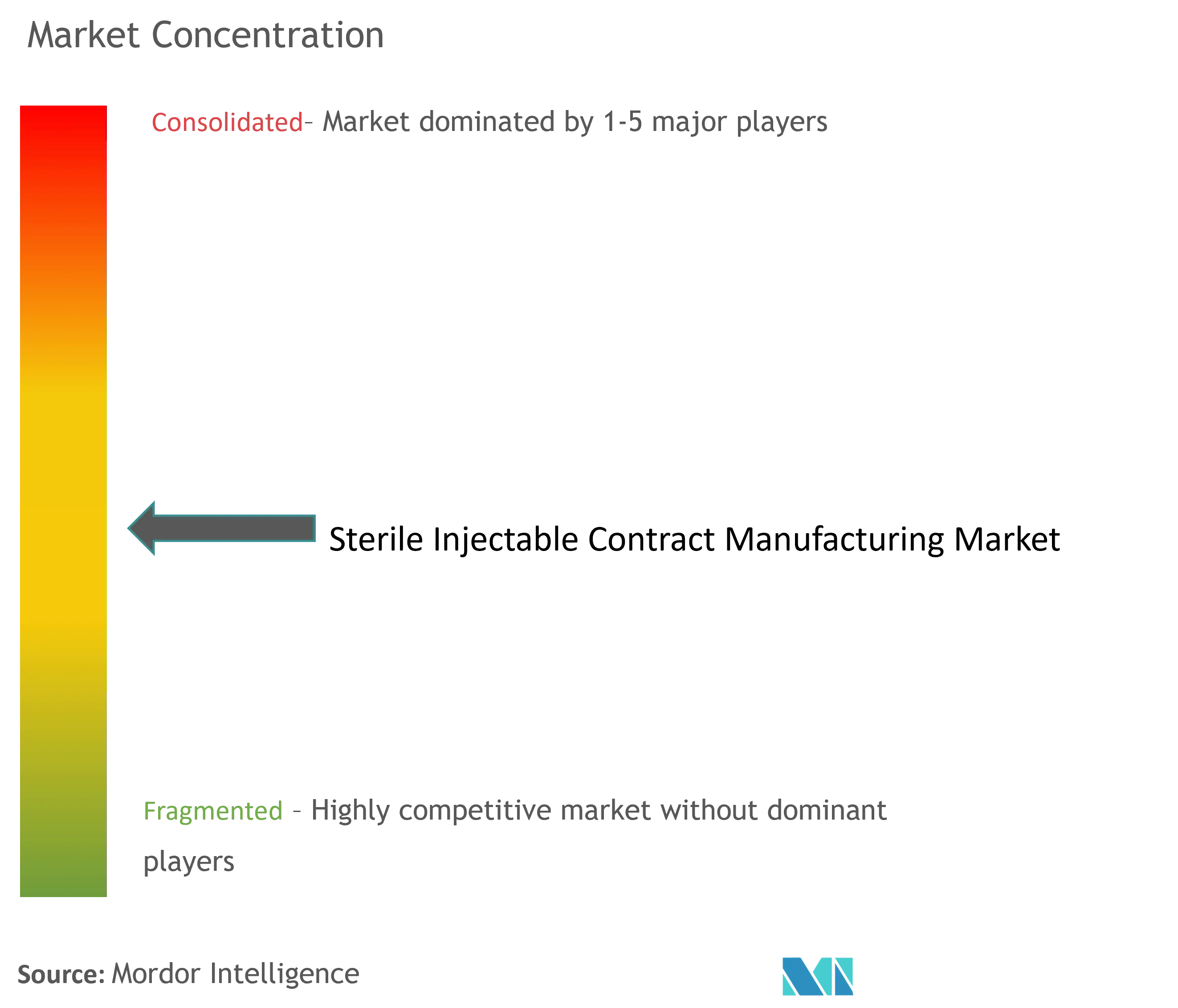
The sterile injectable contract manufacturing market is consolidated and consists of a few major players. In terms of market share, certain major players currently dominate the market. Market players adopt various strategies such as expanding manufacturing facilities, collaborating, and partnering to stay competitive. Some of the companies currently dominating the market are Baxter, Catalent Inc., Vetter Pharma, Recipharm AB, and Aenova Group.
Sterile Injectable Contract Manufacturing Market Leaders
-
Baxter
-
Catalent, Inc
-
Vetter Pharma
-
Recipharm AB
-
Aenova Group
*Disclaimer: Major Players sorted in no particular order
Sterile Injectable Contract Manufacturing Market News
- February 2024: Simtra BioPharma Solutions, a CDMO, announced an investment of more than USD 250 million to expand its sterile fill/finish manufacturing campus in Bloomington, Indiana. The expansion will include a new state-of-the-art 150,000-square-foot building that will feature two high-speed automated isolator syringe fill lines and a new high-speed isolator vial line equipped with three 30-square-meter lyophilizers.
- July 2023: WuXi STA, a contract manufacturing organization, launched its first high potency (HP), fully automated sterile injectable manufacturing line at its drug product site in Wuxi City, China.
Sterile Injectable Contract Manufacturing Market Report - Table of Contents
1. INTRODUCTION
- 1.1 Study Assumptions and Market Definitions
- 1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
- 4.1 Market Overview
-
4.2 Market Drivers
- 4.2.1 Increasing Pipeline and Approvals of Injectables
- 4.2.2 Growing Demand for Biologics and Biosimilars
- 4.2.3 Rise in Investment Across Research and Development Activities for the Development of Novel Therapeutics.
-
4.3 Market Restraints
- 4.3.1 Challenges Related to Quality Control
- 4.3.2 High Operational Costs
-
4.4 Porter's Five Forces Analysis
- 4.4.1 Bargaining Power of Suppliers
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Threat of New Entrants
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value – USD)
-
5.1 By Molecule Type
- 5.1.1 Small Molecule
- 5.1.2 Large Molecule
-
5.2 By Therapeutic Area
- 5.2.1 Cancer
- 5.2.2 Diabetes
- 5.2.3 Cardiovascular Diseases
- 5.2.4 Central Nervous System Diseases
- 5.2.5 Infectious Disorders
- 5.2.6 Musculoskeletal
- 5.2.7 Anti-viral
- 5.2.8 Others
-
5.3 By Route of Administration
- 5.3.1 Subcutaneous (SC)
- 5.3.2 Intravenous (IV)
- 5.3.3 Intramuscular (IM)
- 5.3.4 Others
-
5.4 End User
- 5.4.1 Pharmaceutical and Biopharmaceutical Companies
- 5.4.2 Research Institutes
-
5.5 Geography
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 United Kingdom
- 5.5.2.2 Germany
- 5.5.2.3 France
- 5.5.2.4 Spain
- 5.5.2.5 Italy
- 5.5.2.6 Rest of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 India
- 5.5.3.2 Japan
- 5.5.3.3 China
- 5.5.3.4 Australia
- 5.5.3.5 South Korea
- 5.5.3.6 Rest of Asia-Pacific
- 5.5.4 Middle East and Africa
- 5.5.4.1 GCC
- 5.5.4.2 South Africa
- 5.5.4.3 Rest of the Middle East and Africa
- 5.5.5 South America
- 5.5.5.1 Brazil
- 5.5.5.2 Argentina
- 5.5.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
-
6.1 Company Profiles
- 6.1.1 Baxter
- 6.1.2 Catalent Inc.
- 6.1.3 Vetter Pharma
- 6.1.4 Recipharm AB
- 6.1.5 Aenova Group
- 6.1.6 Fresenius Kabi
- 6.1.7 Unither Pharmaceuticals
- 6.1.8 Famar
- 6.1.9 Cipla Inc.
- 6.1.10 NextPharma Technologies
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
** Subject To AvailablitySterile Injectable Contract Manufacturing Industry Segmentation
As per the scope of the report, sterile injectable contract manufacturing refers to the outsourcing of the production of sterile injectable pharmaceutical products to specialized contract manufacturing organizations (CMOs). CMOs provide services for the manufacturing, filling, and packaging of sterile injectable drugs in compliance with regulatory standards, such as Good Manufacturing Practices (GMP).
The sterile injectable contract manufacturing market is segmented by molecule, therapeutic application, route of administration, end user, and geography. By molecule, the market is segmented into small molecules and large molecules. By therapeutic application, the market is segmented into cancer, diabetes, cardiovascular diseases, central nervous system diseases, infectious disorders, musculoskeletal, anti-viral, and others. By route of administration, the market is segmented into subcutaneous (SC), intravenous (IV), intramuscular (IM), and others. By end user, the market is segmented into pharmaceutical and biopharmaceutical companies and research institutes. By geography, the market is segmented into North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. The report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (USD) for all the above segments.
| By Molecule Type | Small Molecule | |
| Large Molecule | ||
| By Therapeutic Area | Cancer | |
| Diabetes | ||
| Cardiovascular Diseases | ||
| Central Nervous System Diseases | ||
| Infectious Disorders | ||
| Musculoskeletal | ||
| Anti-viral | ||
| Others | ||
| By Route of Administration | Subcutaneous (SC) | |
| Intravenous (IV) | ||
| Intramuscular (IM) | ||
| Others | ||
| End User | Pharmaceutical and Biopharmaceutical Companies | |
| Research Institutes | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Geography | Europe | United Kingdom |
| Germany | ||
| France | ||
| Spain | ||
| Italy | ||
| Rest of Europe | ||
| Geography | Asia-Pacific | India |
| Japan | ||
| China | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Geography | Middle East and Africa | GCC |
| South Africa | ||
| Rest of the Middle East and Africa | ||
| Geography | South America | Brazil |
| Argentina | ||
| Rest of South America |
Sterile Injectable Contract Manufacturing Market Research FAQs
How big is the Sterile Injectable Contract Manufacturing Market?
The Sterile Injectable Contract Manufacturing Market size is expected to reach USD 14.44 billion in 2024 and grow at a CAGR of 12% to reach USD 28.5 billion by 2029.
What is the current Sterile Injectable Contract Manufacturing Market size?
In 2024, the Sterile Injectable Contract Manufacturing Market size is expected to reach USD 14.44 billion.
Who are the key players in Sterile Injectable Contract Manufacturing Market?
Baxter, Catalent, Inc, Vetter Pharma, Recipharm AB and Aenova Group are the major companies operating in the Sterile Injectable Contract Manufacturing Market.
Which is the fastest growing region in Sterile Injectable Contract Manufacturing Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Sterile Injectable Contract Manufacturing Market?
In 2024, the North America accounts for the largest market share in Sterile Injectable Contract Manufacturing Market.
What years does this Sterile Injectable Contract Manufacturing Market cover, and what was the market size in 2023?
In 2023, the Sterile Injectable Contract Manufacturing Market size was estimated at USD 12.71 billion. The report covers the Sterile Injectable Contract Manufacturing Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the Sterile Injectable Contract Manufacturing Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Sterile Injectable Contract Manufacturing Industry Report
Statistics for the 2024 Sterile Injectable Contract Manufacturing market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Sterile Injectable Contract Manufacturing analysis includes a market forecast outlook for 2024 to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.



