United States In Vitro Diagnostics Market Size
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
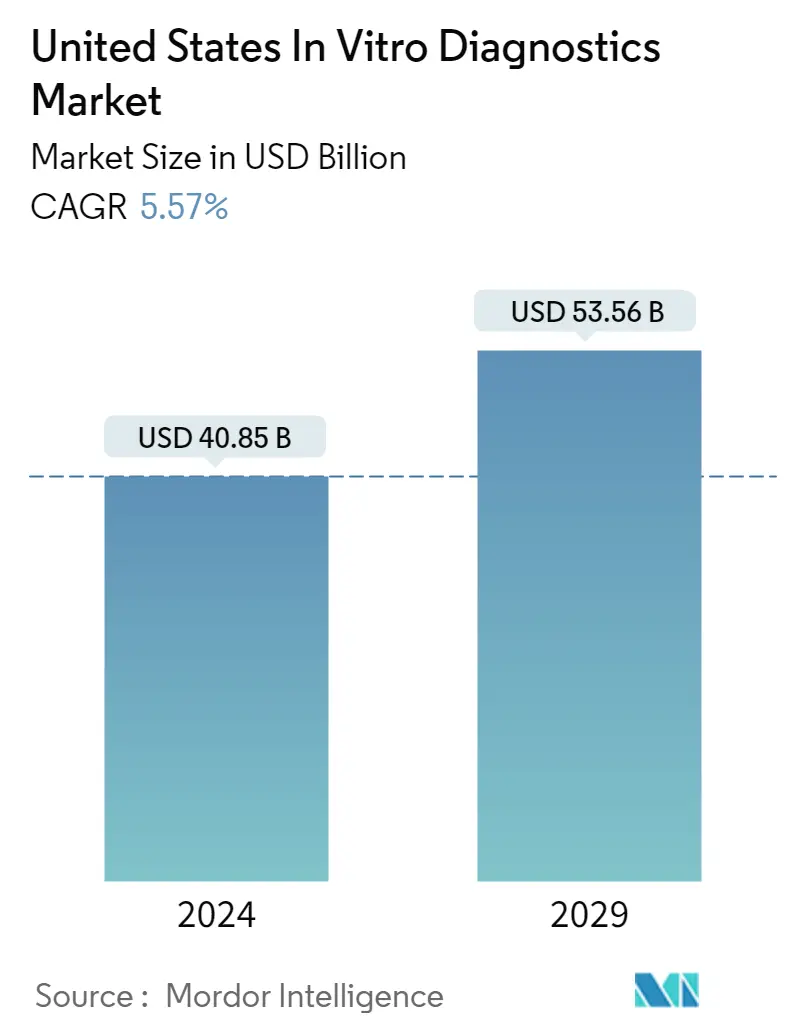
| Market Size (2024) | USD 40.85 Billion |
| Market Size (2029) | USD 53.56 Billion |
| CAGR (2024 - 2029) | 5.57 % |
Major Players
*Disclaimer: Major Players sorted in no particular order |
United States In Vitro Diagnostics Market Analysis
The United States In Vitro Diagnostics Market size is estimated at USD 40.85 billion in 2024, and is expected to reach USD 53.56 billion by 2029, growing at a CAGR of 5.57% during the forecast period (2024-2029).
The COVID-19 pandemic turned the spotlight on in vitro diagnostics since there is an increasing demand for IVD kits and reagents for the rapid and accurate diagnosis of SARS-CoV2 virus infection among the global population. The outbreak of COVID-19 is expected to positively impact the market, as in vitro diagnostics involve the testing of various biological samples. This is expected to aid the diagnosis of infectious diseases, such as COVID-19. Testing remains a crucial step in controlling the COVID-19 pandemic. There has been a shift in the industry dynamics, with the increasing number of players focusing on launching tests for home-based testing. Moreover, in 2021, the United States Food and Drug Administration also prioritized home-based molecular diagnostic tests. In March 2021, BATM Advanced Communications Ltd. announced the launch of its molecular diagnostics self-test kit for the detection of COVID-19. Moreover, these tests enable the early detection of diseases, maintaining a low threat of substitutes. Thus, an increase in demand for newly developed emergency use permitted IVD tests used for COVID-19 detection had a favorable influence on the market.
Key factors propelling the growth of the market are the high prevalence of chronic diseases, increasing the use of POC (Point-of-care) diagnostics, advanced technologies, and increasing awareness, and acceptance of personalized medicines. The increasing government healthcare expenditure and consumer healthcare spending are also responsible for the growth of the market.
According to the United States Census Bureau updates from June 2020, there were more than 54 million people aged 65 and above accounting for around 16.5% of the United States population in 2019. Moreover, it is expected to reach more than 85 million, around 20% of the country's population in 2050. Aging affects the immune system, which increases susceptibility to various diseases. Hence, a large geriatric population requires better healthcare, especially for chronic diseases. The increase in chronic disease cases is expected to increase the demand for in vitro diagnostics, thereby boosting the growth of the market over the forecast period.
Moreover, the increasing product launches with advanced features are expected to drive the market. For instance, in May 2021, Kroger announced the availability of Abbott's BinaxNOW COVID-19 Ag Card for self-testing. This is anticipated to improve the adoption of the product and help manage SARS-CoV-2. Furthermore, in April 2021, Abbott announced the distribution of BinaxNOW COVID-19 Ag Self-Test to retailers, including Walgreens, Walmart, and CVS Pharmacy, in the United States market. Moreover, in January 2020, Quest Diagnostics partnered with Memorial Hermann Health System to provide 21 hospital laboratories in Houston with better, cost-effective, high-quality, and creative diagnostic services.
There are also emerging technological innovations, such as lab-on-a-chip, wearable devices, and POC diagnostics, that are increasingly becoming an important part of the healthcare landscape. These POC diagnostic products have been developed to be used at a patient's bedside in hospitals to get instant results, without the need of sending the samples to the lab. Thus, owing to the ease of use and the ability to provide instant results, the use of POC diagnostics in the United States is increasing rapidly, thereby, boosting the growth of the market studied.
Thus, given the aforementioned factors, the United States in vitro diagnostic market is anticipated to propel over the forecast period.
United States In Vitro Diagnostics Market Trends
This section covers the major market trends shaping the US In Vitro Diagnostics Market according to our research experts:
Cancer/Oncology Segment Segment is Expected to Witness Growth Over the Forecast Period
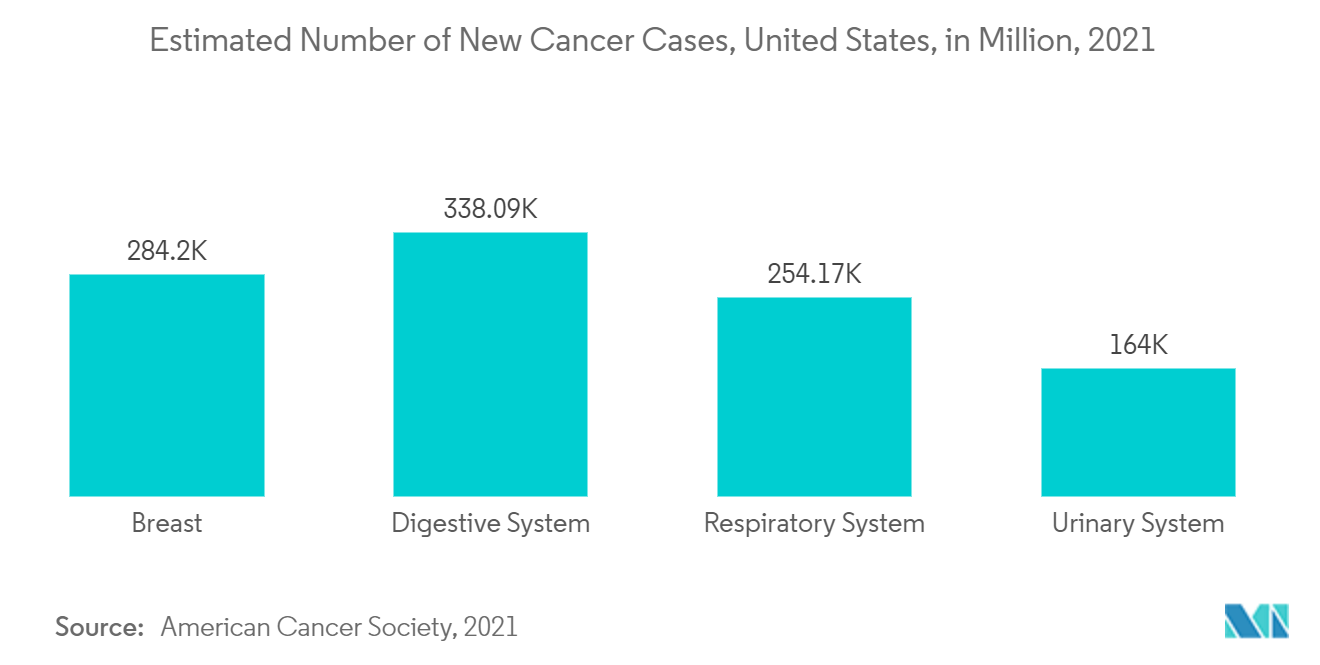
Oncology is expected to witness the fastest CAGR, due to the rising number of cancer cases in the United States. There is also an increasing demand for self-care devices and POC diagnostics in the United States for the treatment of chronic diseases that are expected to boost market growth in the future. According to the American Cancer Society updates from 2021, about 1.8 million new cancer cases were reported in 2020. Breast, prostate, colorectal, lung, stomach, and liver cancers are the most common types. Cancer-causing viruses such as Human Papillomavirus & Hepatitis B Virus/Hepatitis C Virus are also responsible for about 20% of cancer deaths. The number of cancer cases is expected to increase by around 70% over the next two decades, which is expected to increase the demand for clinical testing for early-stage diagnosis. Thus, the adoption of in vitro diagnosis increases with increasing cases of cancer in the region.
Additionally, in August 2022, the United States Food and Drug Administration granted clearance to a blood-based Divitum Tka biomarker assay for disease monitoring of patients with metastatic breast cancer (MBC). Similarly, in November 2020, the Food and Drug Administration approved the liquid biopsy next-generation sequencing-based FoundationOne Liquid CDx test (Foundation Medicine, Inc.) as a companion diagnostic device for multiple additional biomarkers detected in cell free-DNA isolated from plasma specimens.
Moreover, in February 2022, Cancer Moonshot was initiated by the United States government to enhance the screening rate for cancer for identifying the missed cases due to the COVID-19 pandemic. In the next 25 years, the government aims to reduce the cancer death rate by 50% with early diagnosis and treatment.
Thus, the segment is anticipated to witness significant growth over the forecast period due to the abovementioned factors.

United States In Vitro Diagnostics Industry Overview
The In Vitro Diagnostic market is moderately competitive and consists of several major players. Companies, like Abbott Laboratories, Becton, Dickinson and Company, and Danaher Corporation, are adopting strategic actions, mergers, acquisitions, and continued technological advancements, in order to retain their market share in the future. The companies are focusing on increasing technological advancements, in order to address the customer needs, like fully automated systems, that provide fast and accurate results.
United States In Vitro Diagnostics Market Leaders
-
Danaher Corporation
-
bioMerieux SA
-
F Hoffmann-La Roche AG
-
Becton, Dickinson and Company
-
Abbott Laboratories
*Disclaimer: Major Players sorted in no particular order
United States In Vitro Diagnostics Market News
- In July 2022, the United States Food and Drug Administration (FDA) cleared The Lumipulse G β-Amyloid Ratio (1-42/1-40) test, manufactured by Fujirebio Inc., the first diagnostic laboratory test to aid in the diagnosis of Alzheimer's disease.
- In July 2022, Roche announced that the United States Food and Drug Administration (FDA) granted Breakthrough Device Designation to the Elecsys Amyloid Plasma Panel, an innovative new solution to enable Alzheimer's disease to be detected earlier.
United States In Vitro Diagnostics Market Report - Table of Contents
1. INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
- 4.1 Market Overview
-
4.2 Market Drivers
- 4.2.1 High Burden of Chronic Diseases
- 4.2.2 Increasing Use of Point-of-care (POC) Diagnostics Spurring the IVD Market
- 4.2.3 Increasing Government Healthcare Expenditure and Consumer's Healthcare Spending
- 4.2.4 Advanced Technologies Fueling the IVD Market
- 4.2.5 Increasing Awareness and Acceptance of Personalized Medicine and Companion Diagnostics
-
4.3 Market Restraints
- 4.3.1 Lack of Proper Reimbursement
- 4.3.2 Stringent Regulatory Framework
- 4.3.3 Need For High Complexity Testing Centers
-
4.4 Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
-
5.1 By Test Type
- 5.1.1 Clinical Chemistry
- 5.1.2 Molecular Diagnostics
- 5.1.3 Immuno Diagnostics
- 5.1.4 Haematology
- 5.1.5 Other Test Types
-
5.2 By Product
- 5.2.1 Instruments
- 5.2.2 Reagents
- 5.2.3 Other Products
-
5.3 By Usability
- 5.3.1 Disposable IVD
- 5.3.2 Reusable IVD
-
5.4 By Application
- 5.4.1 Infectious Diseases
- 5.4.2 Diabetes
- 5.4.3 Cancer/Oncology
- 5.4.4 Cardiology
- 5.4.5 Autoimmune Diseases
- 5.4.6 Nephrology
- 5.4.7 Other Applications
-
5.5 By End User
- 5.5.1 Diagnostic Laboratories
- 5.5.2 Hospitals and Clinics
- 5.5.3 Other End Users
6. COMPETITIVE LANDSCAPE
-
6.1 Company Profiles
- 6.1.1 Abbott Laboratories
- 6.1.2 F Hoffmann-La Roche AG
- 6.1.3 Becton, Dickinson and Company
- 6.1.4 Danaher Corporation
- 6.1.5 bioMerieux SA
- 6.1.6 Bio-Rad Laboratories Inc.
- 6.1.7 Thermo Fisher Scientific Inc.
- 6.1.8 Qiagen
- 6.1.9 Siemens Healthcare GmbH
- 6.1.10 Sysmex Corporation
- 6.1.11 Arkray Inc.
- 6.1.12 AgilentTechnologies Inc.
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
** Subject To AvailablityUnited States In Vitro Diagnostics Industry Segmentation
As per the scope of the report, in vitro diagnostics are the medical devices and consumables utilized to perform in vitro tests on various biological samples. They are used for the diagnosis of various medical conditions. These in vitro diagnostic products can be instruments, reagents, or any system used for the diagnosis of diseases. The United States In Vitro Diagnostics Market is segmented by Test Type (Clinical Chemistry, Molecular Diagnostics, Immuno Diagnostics, Haematology, and Other Test Types), Product (Instruments, Reagents, and Other Products), Usability (Disposable IVD and Reusable IVD), Application (Infectious Diseases, Diabetes, Cancer/Oncology, Cardiology, Autoimmune Diseases, Nephrology, and Other Applications), and End User (Diagnostic Laboratories, Hospitals and Clinics, and Other End Users). The report offers the value (in USD million) for the above segments.
| By Test Type | Clinical Chemistry |
| Molecular Diagnostics | |
| Immuno Diagnostics | |
| Haematology | |
| Other Test Types | |
| By Product | Instruments |
| Reagents | |
| Other Products | |
| By Usability | Disposable IVD |
| Reusable IVD | |
| By Application | Infectious Diseases |
| Diabetes | |
| Cancer/Oncology | |
| Cardiology | |
| Autoimmune Diseases | |
| Nephrology | |
| Other Applications | |
| By End User | Diagnostic Laboratories |
| Hospitals and Clinics | |
| Other End Users |
United States In Vitro Diagnostics Market Research FAQs
How big is the United States In Vitro Diagnostics Market?
The United States In Vitro Diagnostics Market size is expected to reach USD 40.85 billion in 2024 and grow at a CAGR of 5.57% to reach USD 53.56 billion by 2029.
What is the current United States In Vitro Diagnostics Market size?
In 2024, the United States In Vitro Diagnostics Market size is expected to reach USD 40.85 billion.
Who are the key players in United States In Vitro Diagnostics Market?
Danaher Corporation, bioMerieux SA, F Hoffmann-La Roche AG, Becton, Dickinson and Company and Abbott Laboratories are the major companies operating in the United States In Vitro Diagnostics Market.
What years does this United States In Vitro Diagnostics Market cover, and what was the market size in 2023?
In 2023, the United States In Vitro Diagnostics Market size was estimated at USD 38.69 billion. The report covers the United States In Vitro Diagnostics Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the United States In Vitro Diagnostics Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
United States In Vitro Diagnostics Industry Report
The United States In Vitro Diagnostics market is a dynamic and rapidly evolving sector within the healthcare industry. This market report provides a comprehensive industry analysis, highlighting key market trends and market size. The market segmentation covers various test types, including clinical chemistry, molecular diagnostics, immuno diagnostics, and haematology, among others. The report also delves into the market value of different products such as instruments, reagents, and other related products.
The market forecast offers valuable insights into the expected market growth and market outlook over the coming years. The industry research indicates significant developments in the application of in vitro diagnostics for various diseases, including infectious diseases, diabetes, cancer/oncology, cardiology, autoimmune diseases, and nephrology. The report example available in PDF format provides a detailed market review and industry statistics, essential for understanding the market predictions and market data.
Key industry trends and market leaders are identified, offering a clear view of the competitive landscape. The report also includes a market overview of the end users, which comprises diagnostic laboratories, hospitals and clinics, and other end users. The industry outlook and industry information provided in this report are crucial for stakeholders looking to understand the market dynamics and make informed decisions.
Additionally, the report covers industry sales and industry size, providing a holistic view of the market's current state and future potential. The research companies involved in this study have ensured that the market analysis is thorough, offering a detailed market segmentation and market share insights. The growth rate of the market is also discussed, providing a clear picture of the market's trajectory.
Overall, this market report is an invaluable resource for anyone looking to gain a deep understanding of the United States In Vitro Diagnostics market. The report PDF is available for those who wish to explore the industry reports in greater detail.



